
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
4th Edition
ISBN: 9781119776741
Author: Klein
Publisher: WILEY CONS
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 1, Problem 66ASP
Interpretation Introduction
Interpretation: The correct statement that described the missing formal charge(s) in the following structure should be identified:
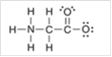
Concept Introduction: Any atom with a formal charge lacks the necessary number of valence electrons. Formal charge is assigned when an atom is present in Lewis structure.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw Lewis structures for tye following molecules.
a.) Atom "X" has 4 valence electrons and Atom "Y" has 6 valence electrons. Draw the Lewis structure for the molecule XY2, showing all lone pairs and formal charges when necessary
b.) Atom "M" has 5 valence electrons and Atom "L" has 7 valence electrons. Draw the Lewis structure for the molecule M2L4, showing all lone pairs and formal charges when necessary
Carbons forms a variety of compounds. Which of the following is NOT a valid reason for this observation?
a. Organic compounds may have many isomers.
b. C can make multiple bonds to O, N, S, or other C.
c. C can form single covalent bonds to O, N, S, and halogens.
d. C forms a total of four covalent bonds.
e. C forms stable covalent bonds to metal atoms.
a. How many shared and unshared electrons are there in the structure?
b. how many shared and unshared pairs of electrons are in the structure?
c. Determine the total number of valence electrons in the structure.
Chapter 1 Solutions
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
Ch. 1.2 - Prob. 1LTSCh. 1.2 - Prob. 2ATSCh. 1.2 - Prob. 2LTSCh. 1.3 - Prob. 3LTSCh. 1.3 - Prob. 4PTSCh. 1.3 - Prob. 5PTSCh. 1.4 - Prob. 4LTSCh. 1.4 - Prob. 7PTSCh. 1.4 - Prob. 8PTSCh. 1.4 - Prob. 9ATS
Ch. 1.5 - Prob. 5LTSCh. 1.5 - Prob. 10PTSCh. 1.5 - Prob. 11ATSCh. 1.5 - Prob. 12ATSCh. 1.6 - Prob. 6LTSCh. 1.6 - Prob. 14ATSCh. 1.7 - Prob. 7LTSCh. 1.7 - Prob. 17ATSCh. 1.10 - Prob. 18CCCh. 1.10 - Prob. 20CCCh. 1.10 - Prob. 8LTSCh. 1.10 - Prob. 21PTSCh. 1.10 - Nemotin is a compound that was first isolated from...Ch. 1.10 - Prob. 23CCCh. 1.11 - Prob. 9LTSCh. 1.11 - Prob. 24PTSCh. 1.11 - Prob. 25PTSCh. 1.11 - Prob. 26PTSCh. 1.11 - Prob. 27ATSCh. 1.12 - Prob. 10LTSCh. 1.12 - Prob. 29ATSCh. 1.13 - Prob. 11LTSCh. 1.13 - Prob. 31ATSCh. 1 - Prob. 32PPCh. 1 - Prob. 33PPCh. 1 - Prob. 34PPCh. 1 - Prob. 35PPCh. 1 - Prob. 36PPCh. 1 - Prob. 37PPCh. 1 - Prob. 38PPCh. 1 - Prob. 39PPCh. 1 - Prob. 40PPCh. 1 - Prob. 41PPCh. 1 - Prob. 42PPCh. 1 - Prob. 44PPCh. 1 - Prob. 45PPCh. 1 - Prob. 46PPCh. 1 - Prob. 47PPCh. 1 - Prob. 48PPCh. 1 - Prob. 49PPCh. 1 - Prob. 50PPCh. 1 - Prob. 51PPCh. 1 - Prob. 52PPCh. 1 - Prob. 53PPCh. 1 - Prob. 54PPCh. 1 - Nicotine is an addictive substance found in...Ch. 1 - Prob. 56PPCh. 1 - Prob. 57PPCh. 1 - Prob. 59PPCh. 1 - Prob. 63ASPCh. 1 - Prob. 64ASPCh. 1 - Prob. 66ASPCh. 1 - Prob. 69ASPCh. 1 - Prob. 71ASPCh. 1 - Prob. 72ASPCh. 1 - Prob. 75IP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 2. Identify the true statement. a. Polar covalent compounds result from a complete transfer of at least one electron. b. Sodium is unlikely to form a bond with lithium. c. Covalent compounds exist in large lattices. d. The smallest unit of an ionic compound is a molecule. 3. A piece of zinc metal is placed in an aqueous solution of magnesium sulfate. The products of this reaction are a. ZnMg(s) + SO4(aq) b. Mg(s) + ZnSO4(aq) c. MgS(s) + ZnO4(aq) d. ZnO(s) + MgSO4(aq) e. There will be no reaction. 4. The shape of the NC13 molecule is a. linear b. trigonal planar c. tetrahedral d. trigonal pyramidal e. V-shaped Imi 5. Which of the following would have the highest boiling point? a. NH3 b. PH3 c. AsH3 d. They would all have approximately the same boiling point. 20 6. Aqueous solutions of silver nitrate and sodium sulfate are mixed. The products of the reaction are a. Ag2SO4(aq) + 2 NaNO3(aq) b. AgSO4(s) + Na2(NO3)2(aq) c. Ag2SO4(s) + 2 NaNO3(aq) d. AgSO4(aq) + Na2(NO3 e. There will be no…arrow_forwardWrite a Lewis structure for each of the following molecules/ions. Be sure to show all non-zero formal charges. Start by counting the valence electrons.arrow_forwardDraw the Lewis structure for CO. A. Calculate the formal charge on each atom in CO. B. Draw the dipole for CO. C. Calculate the oxidation numbers for the carbon and oxygen atoms in CO. D. Formal charges, dipoles, and oxidation numbers are different ways of understanding the charges on covalently-bonded atoms. They tend to agree or be similar. At least in some ways, they disagree for CO. Formal charge guidelines assume that all bonding electrons are shared evenly between the two bonded atoms. Oxidation number guidelines assume that the more electronegative atom in the bond gets all of the electrons. Dipole theory assumes that the bonded electrons are associated more with the more electronegative atom. Now that you understand the basis for the oxidation number guidelines, determine the oxidation number for each carbon in propane assuming that the more electronegative atom gets all of the bonded electrons. You will need to draw the Lewis structure of propane for your answer.arrow_forward
- Choose the INCORRECT statement. In a Lewis structure, the number of valence electrons shown is one more for each negative charge. The central atom is typically the atom with the highest electronegativity. Formal charges are apparent charges associated with atoms in a Lewis structure. Resonance is when more than one plausible structure can be written but the "correct" structure cannot be written.arrow_forwardWhich of the following is the correct guideline in choosing plausible Lewis structure? a. Lewis structure with large formal charges is preferable than with small formal charges b. The negative formal charge is placed on the more electronegative atom c. Lewis structure with many formal charges is preferable d. Lewis structure without formal charge is less plausible than with formal chargesarrow_forwardChemistry: Bonding 4a. Draw the Lewis structure for CH4, (methane), NH3, (ammonia), and H20 (water). b. Calculate the bond polarity for the C-H bond, N-H bond, and O-H bond. c. Classify these molecules as either polar or nonpolar. If the molecule is polar, label the partial negative and partial positive sides of the molecules you drew above.arrow_forward
- Determine if the structural formula below are acceptable Lewis structures for organic compounds. Point out the problems in cases where structure is invalid. CH3 CH;-N-CH-CH3 ČH3 A. This structure is correct because the valance of Nitrogen is complete. B. This is not a correct Lewis structure because Nitrogen can accommodate more atoms. C. This is not a correct Lewis structure because Nitrogen has a charge if it does not have three bonds. D. This is a correct Lewis structure.arrow_forwarda. Which of these three carbon atoms (to the right) has an incomplete octet? b. How many more electrons would be needed to fill each octet? c. What are the formal charges on these carbon atoms?arrow_forwardSteps for Lewis Structures: 1. Determine the total number of valence electrons. Add electrons for negative charges, subtract electrons for positive charges. 2. Place least electronegative atom (except H) as central atom in structure. 3. Connect atoms by singles bonds. Each single bond = 2 electrons. 4. “Sprinkle” remaining electrons around outside atoms first to complete octets. Don’t use more electrons than the total found in step 1. Then complete the central atom’s octet last if you have enough electrons. 5. Make double or triple bonds as needed to complete octets. 6. Place brackets and charge for ions. For the central atom in each formula, draw the Lewis Structure with all valence electrons shown. 1. PH3 2. H2O 3. CO2 4. CHCl3 5. O2 6. N2 7. CF4 8. C3H8 9. CH3COOH 10. N2O 11. OCN-arrow_forward
- Identify if the statement is true or false. If false, explain why the statement is false. a. H for the formation of a bond is always a negative number. b. Because the presence of pi bonds does not influence the geometry of a molecule, the presence of pi bonds does not affect the value of the bond enthalpy between two atoms. c. A hot metal is placed in a calorimeter. The temperature of the water increases. The q value for the metal will be positive and the q value for the water will be negative.arrow_forward1. How many bonds must each of the following atoms have in order to have no formal charge? a. Carbon b. Hydrogen c. Oxygen d. Chlorine e. Nitrogen 2. Using the information from question #1, determine whether each of the following structures is possible or impossible? (Note that if no formal charges are shown, this implies that all formal charges are zero.) H H H H I MacBook Pro H Harrow_forwardWhich structure is formed when there are five valence shell electron pairs and four are bonded pairs whereas one pair is a lone pair? A. Linear B. T- Shape C. Trigonal bipyramid D. Seesawarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License