
Concept explainers
Interpretation: Whether the answers in CTQ are in agreement with Memorization Task NW1.2 given below or not should be given.
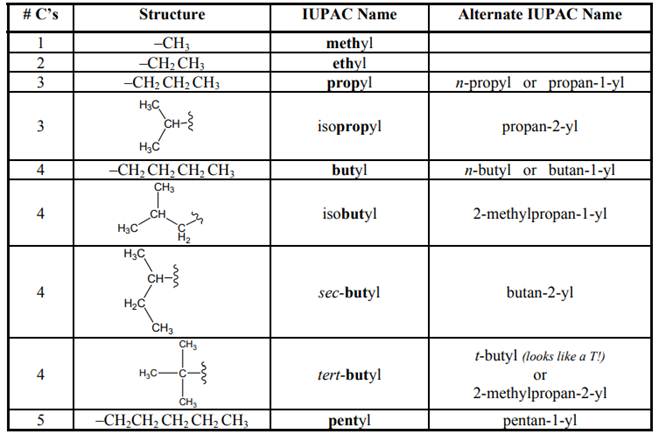
Concept introduction: Systematic way to name different organic compounds is
Rules for nomenclature of
1 The longest continuous carbon chain is identified first and named in accordance with number of carbon atoms present in it. For example, hydrocarbon with one carbon atom has prefix “meth”, that with two carbon atoms has prefix “eth”, that with three carbon atoms has prefix “prop” and so on. Suffix used for alkanes is “ane.”
2. Substituents attached to parent carbon chain are to be identified. These are named by removal of single hydrogen atom from carbon chain end and named by replacement of suffix “ane” by “yl.” For example if
3. Carbons of parent chain are named in such way that substituents acquire the lowest numbers.
4. If same substituent is present more than one time in molecule, it is represented by prefix “di”, “tri” and so on. It depends on number of times substituent occurs in molecule.
5. If two or more substituents are present in molecule, these are named in alphabetical order.
6. If carbon chains of same length exist in same molecule, chain with the largest number of side chains, followed by lowest number to substituents, chain with the greatest number of carbon atoms in smaller chain and chain with the least branched side chains are preferred over other ones.
7. Prefix “cyclo” is used if cyclic alkane is present in molecule.
Want to see the full answer?
Check out a sample textbook solution
Chapter NW1 Solutions
Organic Chemistry: A Guided Inquiry
- What is the missing reactant in this organic reaction? || CH3-C- CH2 -NH2 R No Answer A CH3 CH3-N-CH₂ || C−NH–CH2 C-CH3 + H₂O Specifically, in the drawing area below draw the structure of R. If there is more than one reasonable answer, you can draw any one of them. If there is no reasonable answer, check the No answer box under the drawing area. Note for advanced students: you may assume no products other than those shown above are formed. Click and drag to start drawing a structure. || X :0 Śarrow_forwardOrganic Functional Groups Identifying positions labeled with Greek letters in acids and derivatives If possible, replace an H atom on the a carbon of the molecule in the drawing area with a methyl group substituent, and replace an H atom on the ß carbon with a hydroxyl group substituent. If one of the substituents can't be added for any reason, just don't add it. If neither substituent can be added, check the box under the drawing area. HO. Oneither substituent can be added. D X 1/5 0:0 Tyler V ola Ararrow_forwardOrganic Functional Groups Identifying positions labeled with Greek letters in acids and derivatives If possible, replace an H atom on the a carbon of the molecule in the drawing area with a methyl group substituent, and replace an H atom on the ß carbon with a hydroxyl group substituent. If one of the substituents can't be added for any reason, just don't add it. If neither substituent can be added, check the box under the drawing area. HO Oneither substituent can be added. OH 0/5 Xarrow_forward
- (CQ9) Hello! Please help me answer this one. Please refer to the given picture/s below for the questions. Please read the instructions and directions very carefully. Double and triple check your answers, previous tutors got it wrong. NOTE: Type only your answers. Please do not handwritten your answers. Make sure your formulas, solutions and answers' format are all correct. Make sure your diagram isn't handwritten. I want a typewritten only!arrow_forwardUse the IUPAC Nomenclature System to name the following ester: CH₂ | CH,—CH–CH,-C-O-CH, X 5arrow_forward(E) What suffix do all the names in Model 1 have in common with each other?arrow_forward
- Use Model 1 to propose names for three-, four-, five-, and six-carbon branches that follow the same pattern as “methyl” and “ethyl” for one- and two-carbon branches, respectively. (Note: The names of seven-, eight-, etc. carbon branches follow the same pattern, but branches of such length are rare since they are usually the parent chain.)arrow_forward3. Use curved arrows to show electron movement in the reactant side and draw the product/s of the Lewis (nucleophile-electrophile) reaction. Draw in all lone pairs and charges where appropriate. acid-base -co +arrow_forwardModified True or False. Write correct if the statement is True and if false write the word/s that make it false and beside it write the word/s that will make the statement true. 4. Lone pair delocalization decreases the positivity of carbonyl carbon.5. The longer the carbon chain, the higher the boiling point.6. sp3 is 75% p character and this allows C-H hyper conjugation.7. The higher the electronegativity, the higher the temperature needed to break bonds.8. Steric effect increases the boiling point.9. Stearic effect increases the Van der Waals forces.10. Lone pair delocalization decreases the positivity of carbonyl carbon.11. Amines are considered basic.12. The stronger the Van der Waals forces between molecules of the same substance, themore soluble the substance is in water.13. The longer the carbon chain, the lower the solubility of the substance in water.14. Alkanes with less than 6 carbon atoms are gases.15. Stearic effects enhances the solubility in water.16. H-bond can exist…arrow_forward
- Pls help ASAP. Pls do all asked questions. Pls name this compound.arrow_forward7. Draw curved arrows indicating the movements of electrons between the following pair of resonance structures. Name the pattern of resonance shown. What is the hybridization of the carbon atoms? 8. Draw the remaining three resonance structures for the molecule in problem 7 above. 9. There are several possible forms of a trisubstituted cyclohexane with the formula C10H200. I have drawn four of them. From these, which one do you think is most commonly naturally occurring, and why? Which is least commonly occurring and why? HO HO" HO HOarrow_forwardIV) Biomolecules - Below are examples of three classes of biomolecules we will not discuss in this class: OH What is the R/S configuration of the indicated carbon? H. Circle one: More soluble in. Water Нехane HO Circle the most acidic proton (if it is not shown, draw it in and circle it). estrogen (lipid) NH2 How many stereocenters does this tripeptide have? Circle one: Is the circled functional group aromatic? YES NO H2N What is the Index of Hydrogen Deficiency of this peptide? NH A tripeptide (three amino acids) Name the circled functional group: What is the hybridization of the indicated oxygen? (CH2)16CH3 Circle one: Is this triacylglyceride optically active? YES NO H3C(H2C)16' Circle one: More soluble in... Water Oil -(CH2)16CH3 Explain why the triacylglyceride is more soluble in your choice above: a triacylglyceridearrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
