
Concept explainers
The hydroxyl radical
(d) The radical is generated when sunlight hits water vapor. Calculate the maximum wavelength (in nm) required to break an
Interpretation:
The Lewis structure of hydroxyl radical is to be drawn; the reason for radical’s high affinity for H atoms, enthalpy change for the given reaction, and the maximum wavelength required to break given bond in given compound are to be calculated.
Concept introduction:
The standard enthalpy of a reaction is the enthalpy change that occurs under standard conditions.
The standard enthalpy of a reaction is determined using the equation as given below:
Here, the stoichiometric coefficients are represented by m for reactants and n for products, and enthalpy of formation at standard conditions is represented by
The standard enthalpy of formation is the amount of heat change that arises as a result of one mole of compound formation at the standard states from its integral elements.
The value of enthalpy of formation of an element is zero at its most stable state.
Bond Enthalpy is the energy required to break a bond of 1 mole of a substance. It is given in
The Lewis dot symbols contain dots, which give information about valence electrons.
According to Planck’s energy–frequency law, energy is given by:
Here, h is Planck’s constant,
Answer to Problem 146AP
Solution:
(a)
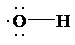
(b) The reason for radical
(c)
(d)
Explanation of Solution
a) Lewis structure for radical (OH)
The skeletal structure for
The number of valence electrons is as follows:
Now, only two electrons are used in bond formation. So, the number of remaining electrons is 5.
The oxygen atom has only one electron used in bond formation, so it will have 5 remaining electrons.
So, the Lewis structure for
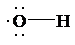
b) The radical has high affinity for H atom, refer table 8.6.
From table 8.6, the bond enthalpy of oxygen–hydrogen bond:
Since the value of oxygen–hydrogen bond is very high, the radical
Hence, the reason for radical
c) The enthalpy change for the following given reaction:
The enthalpy of reaction can be calculated as follows:
From table 8.6, the enthalpy of formation values are as follows:
Now, the standard enthalpy of the given reaction is as follows:
Substitute
Hence, the enthalpy of the given reaction from the bond enthalpy values is
d) The maximum wavelength (in nm) required to break an
From table 8.6, the bond enthalpy for oxygen–hydrogen bond is as follows:
This can be written as the energy of oxygen–hydrogen bond as
According to Planck’s energy–frequency law, energy is given as follows:
This can be written as follows:
Substitute
Hence, the maximum wavelength required to break the oxygen–hydrogen bond is
Want to see more full solutions like this?
Chapter 8 Solutions
Chemistry
- Bond Enthalpy When atoms of the hypothetical element X are placed together, they rapidly undergo reaction to form the X2 molecule: X(g)+X(g)X2(g) a Would you predict that this reaction is exothermic or endothermic? Explain. b Is the bond enthalpy of X2 a positive or a negative quantity? Why? c Suppose H for the reaction is 500 kJ/mol. Estimate the bond enthalpy of the X2 molecule. d Another hypothetical molecular compound, Y2(g), has a bond enthalpy of 750 kJ/mol, and the molecular compound XY(g) has a bond enthalpy of 1500 kJ/mol. Using bond enthalpy information, calculate H for the following reaction. X2(g)+Y2(g)2XY(g) e Given the following information, as well as the information previously presented, predict whether or not the hypothetical ionic compound AX is likely to form. In this compound, A forms the A+ cation, and X forms the X anion. Be sure to justify your answer. Reaction: A(g)+12X2(g)AX(s)The first ionization energy of A(g) is 400 kJ/mol. The electron affinity of X(g) is 525 kJ/mol. The lattice energy of AX(s) is 100 kJ/mol. f If you predicted that no ionic compound would form from the reaction in Part e, what minimum amount of AX(s) lattice energy might lead to compound formation?arrow_forwardhat is the enthalpy change for a process? Is enthalpy a state function? In what experimental apparatus are enthalpy changes measured?arrow_forwardWrite all resonance structures of chlorobenzene, C6H5Cl, a molecule with the same cyclic structure as benzene. In all structures, keep the CCl bond as a single bond. Which resonance structures are the most important?arrow_forward
- Using the bond dissociation enthalpies in Table 8.8, estimate the enthalpy of combustion of gaseous methane, CH4, to give water vapor and carbon dioxide gas.arrow_forwardNitrosyl azide, N4O, is a pale yellow solid first synthesized in 1993. Write the Lewis structure for nitrosyl azide.arrow_forward7.18 In terms of the strengths of the covalent bonds involved, why do combustion reactions release energy?arrow_forward
- In which of the following molecules does the sulfur have an expanded octet? For those that do, write the Lewis structure. (a) SO2 (b) SF4 (c) SO2Cl2 (d) SF6arrow_forwardExplain how bond energies can be used to estimate E for a reaction. Why is this an estimate of E? How do the product bond strengths compare to the reactant bond strengths for a reaction that releases energy? A reaction that gains energy? What is the relationship between the number of bonds between two atoms and bond strength? Bond length?arrow_forwardExplain the decomposition of nitroglycerin in terms of relative bond enthalpies.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





