
Concept explainers
(a)
Interpretation:
Structural formula for N,N-diethylpropanamide has to be drawn.
Concept Introduction:
Structure of the amide can be drawn from the IUPAC name. In the IUPAC name, the parent chain of carbon atom can be identified and then the substituents present in it can also be identified. With this information, the structure for the given compound can be drawn. In an amide the counting has to be always from the carbonyl carbon that is given the number 1.
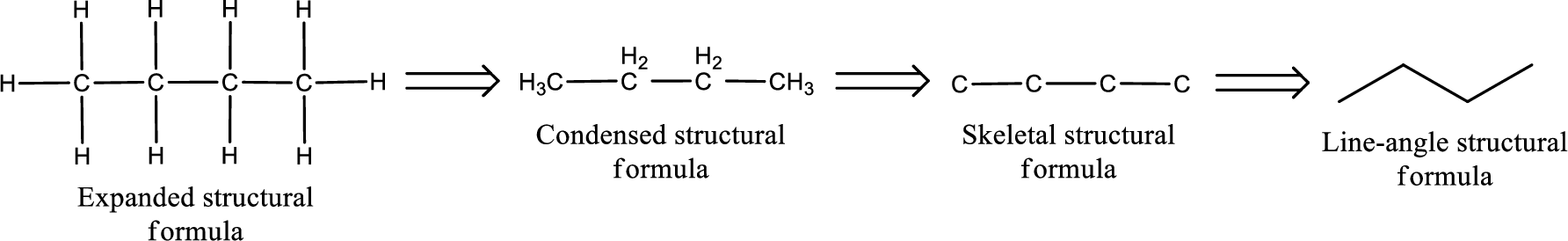
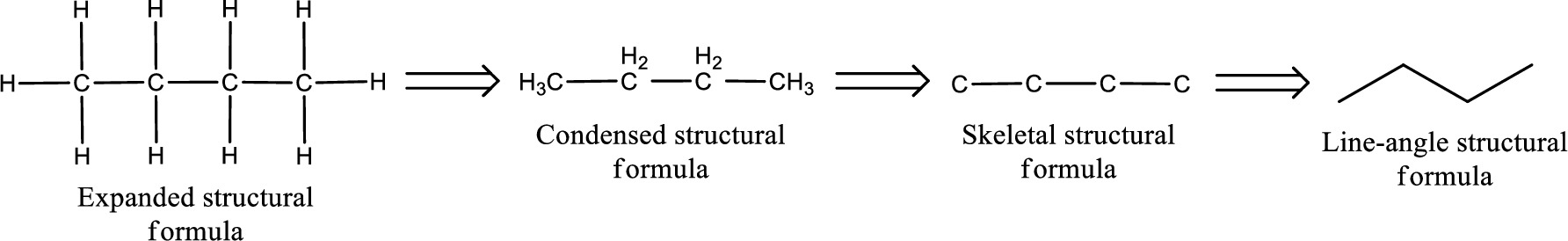
The structural representation of organic compound can be done in 2D and 3D. In two-dimensional representation, there are four types of representation in which an organic compound can be drawn. They are,
- Expanded structural formula
- Condensed structural formula
- Skeletal structural formula
- Line-angle structural formula
Structural formula which shows all the atoms in a molecule along with all the bonds that is connecting the atoms present in the molecule is known as Expanded structural formula.
Structural formula in which grouping of atoms are done and in which the central atoms along with the other atoms are connected to them are treated as group is known as Condensed structural formula.
Structural formula that shows the bonding between carbon atoms alone in the molecule ignoring the hydrogen atoms being shown explicitly is known as Skeletal structural formula.
Structural formula where a line represent carbon‑carbon bond and the carbon atom is considered to be present in each point and the end of lines is known as Line-angle structural formula.
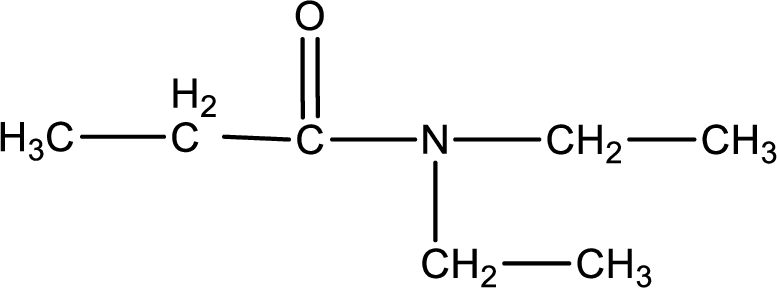
(a)
Answer to Problem 6.114EP
The structural formula for N,N-diethylpropanamide is,
Explanation of Solution
The given name of the compound is N,N-diethylpropanamide. From the name it is understood that the parent carbon chain is propane and it contains three carbon atoms. The parent chain can be drawn as shown below,

As the given compound is an amide, one of the carbon atoms has to be carbonyl group and a nitrogen atom has to be attached to the carbonyl carbon atom.
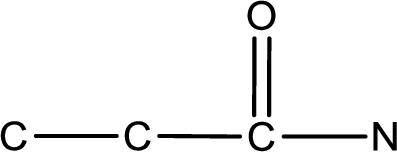
The substituents present in the given name are two ethyl groups on the nitrogen atom. This gives the structure of the given compound as shown below,
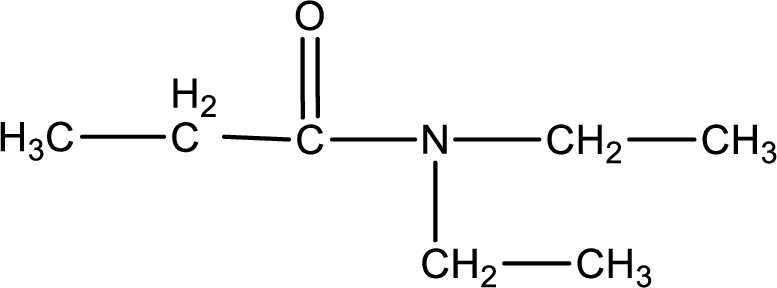
Structural formula for the given N,N-diethylpropanamide is drawn.
(b)
Interpretation:
Structural formula for
Concept Introduction:
Structure of the amide can be drawn from the IUPAC name. In the IUPAC name, the parent chain of carbon atom can be identified and then the substituents present in it can also be identified. With these information, the structure for the given compound can be drawn. In an amide the counting has to be always from the carbonyl carbon that is given the number 1.
The structural representation of organic compound can be done in 2D and 3D. In two-dimensional representation, there are four types of representation in which an organic compound can be drawn. They are,
- Expanded structural formula
- Condensed structural formula
- Skeletal structural formula
- Line-angle structural formula
Structural formula which shows all the atoms in a molecule along with all the bonds that is connecting the atoms present in the molecule is known as Expanded structural formula.
Structural formula in which grouping of atoms are done and in which the central atoms along with the other atoms are connected to them are treated as group is known as Condensed structural formula.
Structural formula that shows the bonding between carbon atoms alone in the molecule ignoring the hydrogen atoms being shown explicitly is known as Skeletal structural formula.
Structural formula where a line represent carbon‑carbon bond and the carbon atom is considered to be present in each point and the end of lines is known as Line-angle structural formula.
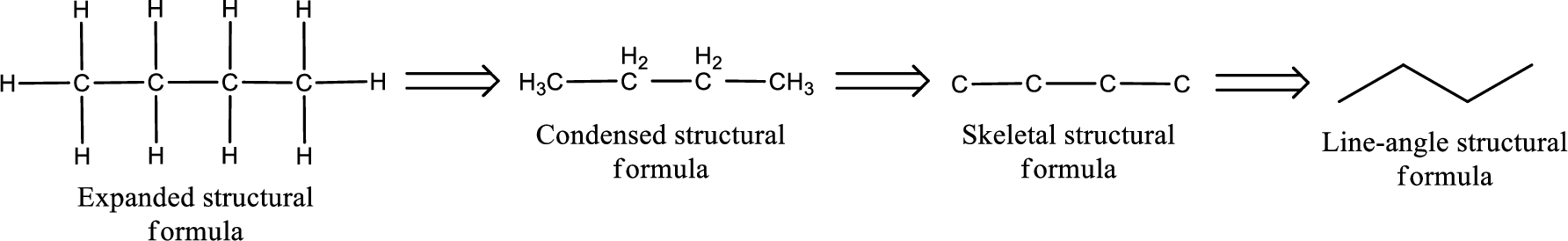
(b)
Answer to Problem 6.114EP
The structural formula for
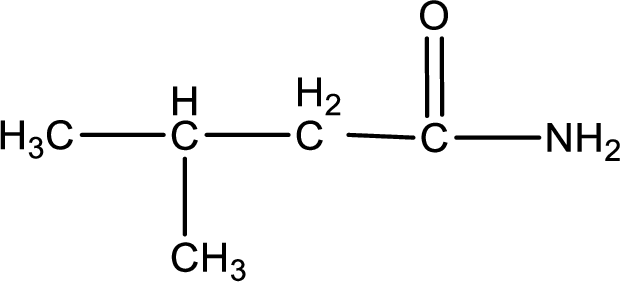
Explanation of Solution
The given name of the compound is
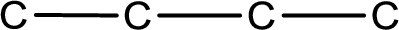
As the given compound is an amide, one of the carbon atom has to be carbonyl group and a nitrogen atom has to be attached to the carbonyl carbon atom.
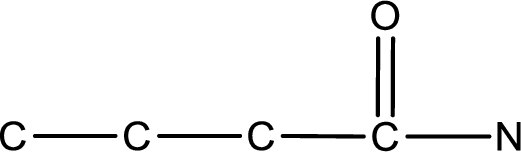
The substituent present in the given name is a methyl groups on the beta carbon atom. This gives the structure of the given compound as shown below,
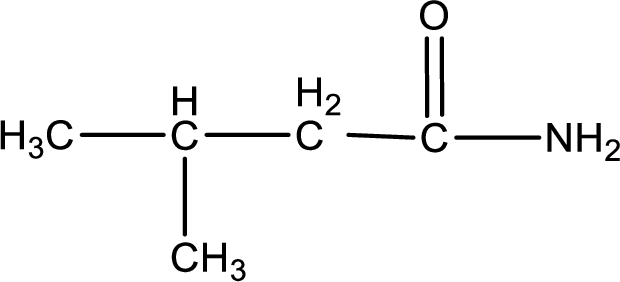
Structural formula for the given
(c)
Interpretation:
Structural formula for N-methylbenzamide has to be drawn.
Concept Introduction:
Structure of the amide can be drawn from the IUPAC name. In the IUPAC name, the parent chain of carbon atom can be identified and then the substituents present in it can also be identified. With these information, the structure for the given compound can be drawn. In an amide the counting has to be always from the carbonyl carbon that is given the number 1.
The structural representation of organic compound can be done in 2D and 3D. In two-dimensional representation, there are four types of representation in which an organic compound can be drawn. They are,
- Expanded structural formula
- Condensed structural formula
- Skeletal structural formula
- Line-angle structural formula
Structural formula which shows all the atoms in a molecule along with all the bonds that is connecting the atoms present in the molecule is known as Expanded structural formula.
Structural formula in which grouping of atoms are done and in which the central atoms along with the other atoms are connected to them are treated as group is known as Condensed structural formula.
Structural formula that shows the bonding between carbon atoms alone in the molecule ignoring the hydrogen atoms being shown explicitly is known as Skeletal structural formula.
Structural formula where a line represent carbon‑carbon bond and the carbon atom is considered to be present in each point and the end of lines is known as Line-angle structural formula.
(c)
Answer to Problem 6.114EP
The structural formula for N-methylbenzamide is,
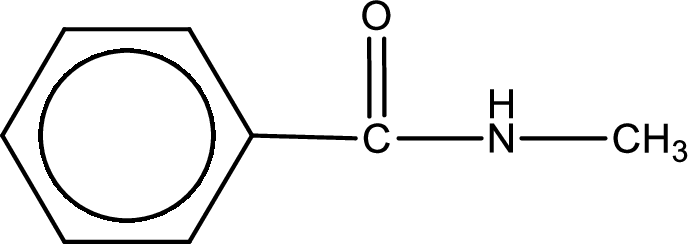
Explanation of Solution
The given name of the compound is N-methylbenzamide. From the name it is understood that the parent carbon chain is benzene and it contains six carbon atoms. The parent chain can be drawn as shown below,
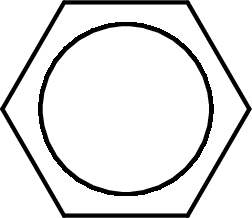
The name itself says that an amide group is attached to a benzene ring. On the nitrogen atom of the amide group, a methyl group is present as substituent.
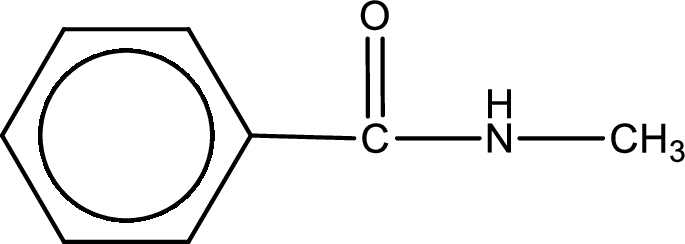
Structural formula for the given N-methylbenzamide is drawn.
(d)
Interpretation:
Structural formula for
Concept Introduction:
Structure of the amide can be drawn from the IUPAC name. In the IUPAC name, the parent chain of carbon atom can be identified and then the substituents present in it can also be identified. With these information, the structure for the given compound can be drawn. In an amide the counting has to be always from the carbonyl carbon that is given the number 1.
The structural representation of organic compound can be done in 2D and 3D. In two-dimensional representation, there are four types of representation in which an organic compound can be drawn. They are,
- Expanded structural formula
- Condensed structural formula
- Skeletal structural formula
- Line-angle structural formula
Structural formula which shows all the atoms in a molecule along with all the bonds that is connecting the atoms present in the molecule is known as Expanded structural formula.
Structural formula in which grouping of atoms are done and in which the central atoms along with the other atoms are connected to them are treated as group is known as Condensed structural formula.
Structural formula that shows the bonding between carbon atoms alone in the molecule ignoring the hydrogen atoms being shown explicitly is known as Skeletal structural formula.
Structural formula where a line represent carbon‑carbon bond and the carbon atom is considered to be present in each point and the end of lines is known as Line-angle structural formula.
(d)
Answer to Problem 6.114EP
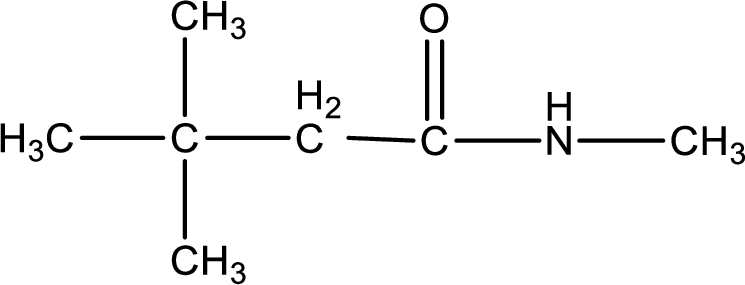
The structural formula for
Explanation of Solution
The given name of the compound is
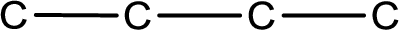
As the given compound is an amide, one of the carbon atom has to be carbonyl group and a nitrogen atom has to be attached to the carbonyl carbon atom.
The substituents present in the given name are three methyl groups. Out of this two on beta carbon atom and one on nitrogen atom. This gives the structure of the given compound as shown below,
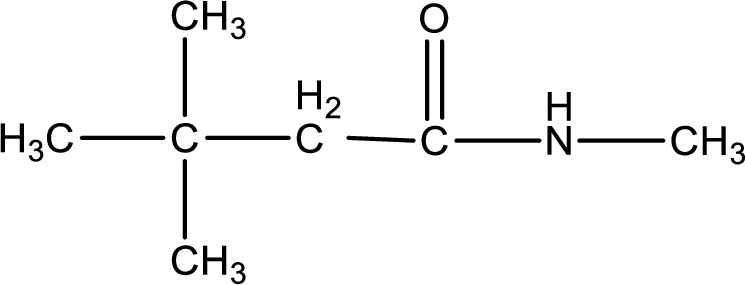
Structural formula for the given
Want to see more full solutions like this?
Chapter 6 Solutions
Organic And Biological Chemistry
- N-p-hydroxyphenylethanamide is commonly known as a. acetaminophen b. acetamide c. acetanilide d. formamide High molar mass amines have __________ odor. a.strong ammoniacal b.fruity c.fishy d.obnoxious Trimethyl amine has _________ odor. a.obnoxious b.fishy c. ammoniacal d. fruityarrow_forwardUse the structure of aniline provided and draw the condensedformula for each of the following amines.a. N-Methylaniline c. N-Ethylanilineb. N,N-Dimethylaniline d. N-Isopropylanilinearrow_forwarda.Primary amides tend to exist as dimers in the solid and liquid state. b.Dimethylacetamide, CH3CON(CH3)2 has a higher boiling point than acetamide. c.Nitrile is often classified as an acid derivative because it is hydrolyzed to a -COOH. d.Esters have lower boiling points than ketones of comparable molecular masses. Whice are correct?arrow_forward
- Amide hydrolysis in basic conditions forms A. a carboxylic acid and an amine B. a carboxylate salt and an amine 3. an ester and an amine 4. a carboxylic acid and an amine saltarrow_forwardWhich of the following statements is true for an amine if "N-" is part of the IUPAC name? a. The compound is a primary amine. b. The molecule is contains a nitrogen atom attached to carbon number one. c. The compound is a secondary amine. d. The compound is a tertiary amine.arrow_forwardanswer all questions / partsarrow_forward
- Question 16.arrow_forwardanswer all questions / partsarrow_forwardWhich of the following is a suitable name for the ester CH3CH2CH2CH2CO2CH2CH2CH2CH3 Select one: a. pentyl butanoate b. nonanoate c. butyl pentanoate d. pentanebutanate Amides have Select one: a. an -OR group bonded to the carbonyl C atom b. an -NH2, -NHR, or NR2 group bonded to the carbonyl C atom c. an -OH group bonded to the carbonyl C atomarrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





