
Concept explainers
Interpretation:
The two product in the given reaction are formed after [1, 7] sigmatropic rearrangement, one due to hydrogen migration and the other to deuterium migration. It should be represented the configuration of product by replacing the A and B with appropriate atoms (H or D)
Concept introduction:
In a sigmatropic reaction “ one new sigma-bond is formed as another breaks.”
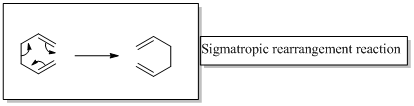
Sigma tropic rearrangement reactions are designated with digits. For example a [1, 3] sigma tropic rearrangement describe a reaction in which the residue migrates from position 1 to position 3
Woodward –Hoffmann rules are the set of rules used to vindicate or predict certain aspects of the stereo chemical outcome and activation energy of pericyclic reactions.
Woodward – Hoffmann rules for sigma tropic rearrangement reactions are listed below
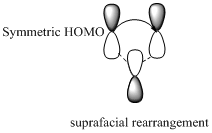
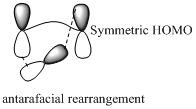
Migration of carbon and hydrogen will occur in a sigmatropic rearrangement reaction. Migration of hydrogen in suprafacial and antarafacial rearrangement can be represented as follows,


Carbon migrating with one lobe of its p orbital interacting

Carbon migrating with both lobe of its p orbital interacting

Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
Organic Chemistry (8th Edition)
- (1R,2R)-1-Bromo-2-methylcyclopentane is reacted with sodium methoxide. Given the product(s) and show the reaction mechanism, including the depiction of the transition state. Draw an energy diagram for this conversion.arrow_forwardCompounds X and Y are both C7H15Cl products formed in the radical chlorination of 2,4-dimethylpentane. Base-promoted E2 elimination of X and Y gives, in each case, a single C7H₁4 alkene. Both X and Y undergo an SN2 reaction with sodium iodide in acetone solution to give C7H15l products; in this reaction Y reacts faster than X. What is the structure of X? • Do not use stereobonds in your answer. • In cases where there is more than one possible structure for each molecule, just give one for each. . Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate structures with + signs from the drop-down menu. наarrow_forwardIn a strongly acidic solution, cyclohexa-1,4-diene tautomerizes to cyclohexa-1,3-diene.Propose a mechanism for this rearrangement, and explain why it is energetically favorablearrow_forward
- 7. Provide the synthesis for the following product starting with the provide starting material. More than one step is required. Show the mechanism for each reaction. CI- Cl- Starting Material Productarrow_forwardMake a table with the main conditions for each mechanism: E1, E2, SN1, SN2.arrow_forwardYour task is to convert 2-bromobutane to 1-butene in highest yield. Which reagents would you use? O KOH/H2O O KOH/CH,OH O CH3CH2ONA/CH3CH2OH O CH3ONA/CH3OH O (CH3)3COK/(CH3)3COHarrow_forward
- Compounds X and Y are both C,H15CI products formed in the radical chlorination of 2,4-dimethylpentane. Base-promoted E2 elimination of X and Y gives, in each case, a single C-H14 alkene. Both X and Y undergo an SN2 reaction with sodium iodide in acetone solution to give C-H151 products; in this reaction Y reacts faster than X. What is the structure of X? • Do not use stereobonds in your answer. • In cases where there is more than one possible structure for each molecule, just give one for each. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate structures with + signs from the drop-down menu. In (F ChemDoodlearrow_forwardA student adds NBS to a solution of 1-methylcyclohexene and irradiates the mixture with a sunlamp until all the NBS has reacted. After a careful distillation, the product mixture contains two major products of formula C7H11Br. (a) Draw the resonance forms of the three possible allylic free radical intermediates.arrow_forwardPredict the mechanism(s) (SN1, SN2, E1, E2) and major product(s) for each reaction.arrow_forward
- Propose a mechanism for the following cycloaddition reaction. Classify the reaction using the [m + n] classifications for cycloadditions. Draw the bonding molecular orbitals and identify the type of bonding present (supra - supra, antaraantara, or supra - antara). Identify if this reaction would proceed thermally or photochemically.arrow_forward2. The following reaction results in two possible carbocation rearrangement products, each of which can be characterized by a change in the ring size. Give structures for both products. There is no need to show the mechanism of the reaction. HCI (conc)arrow_forwardConsider the following reaction. [1] Would this reaction proceed through a SN1, Sn2, E1 or E2 mechanism to produce the product shown? [2] Provide a detailed step-wise mechanism for the formation of the product. Provide all steps, intermediates, formal charges, and show the movement of electrons with curved arrows. [3] Identify the arrow pushing pattern (mechanistic pattern) for each step. [4] Draw a transition state for each step. H20 Br + enarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





