
Organic Chemistry (8th Edition)
8th Edition
ISBN: 9780134042282
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 28, Problem 42P
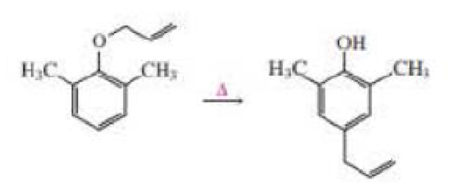
Propose a mechanism for the following reaction:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
a) Draw the octahedral mer-[FeCl3(CN)3] complex and determine its point group. Use proper wedges and
dashes in order to illustrate 3 dimensional details. Use the point group to determine if the complex has a
resulting net dipole moment and describe its allowed direction with respect to its symmetry elements (if
applicable).
ード
M
4-
b) Substitute one chlorido ligand in mer-[FeCl3(CN)3] 4 with one fluorido ligand. Determine all possible
isomers and their corresponding point groups. Use the point groups to determine if the complexes have
resulting net dipole moments and describe their allowed direction with respect to its symmetry elements (if
applicable). The number of complex sketches below is not necessarily indicative of the number of isomers.
4-
4-
☐☐☐
c) Substitute two chlorido ligands in mer-[FeCl3 (CN)3] 4 with two fluorido ligands. Determine all possible
isomers and their corresponding point groups.. Use the point groups to determine if the complexes have
resulting net dipole…
Show work. don't give Ai generated solution
Differentiate electron spin and electron spin moment.
Chapter 28 Solutions
Organic Chemistry (8th Edition)
Ch. 28.1 - Prob. 1PCh. 28.2 - Prob. 2PCh. 28.2 - Prob. 3PCh. 28.2 - Give a molecular orbital description for each of...Ch. 28.3 - Prob. 5PCh. 28.3 - Prob. 6PCh. 28.3 - Prob. 7PCh. 28.3 - Prob. 8PCh. 28.4 - Prob. 10PCh. 28.4 - Prob. 11P
Ch. 28.5 - Prob. 12PCh. 28.5 - a. Draw the product of the following reaction: b....Ch. 28.5 - Prob. 14PCh. 28.5 - Prob. 15PCh. 28.5 - Prob. 17PCh. 28.5 - Prob. 18PCh. 28.6 - Prob. 19PCh. 28.6 - Explain why the hydrogen and the methyl...Ch. 28.6 - Chorismate mutase is an enzyme that promotes a...Ch. 28.7 - Convince yourself that the TE-AC method for...Ch. 28 - Draw the product of each of the following...Ch. 28 - Draw the product of each of the following...Ch. 28 - Prob. 25PCh. 28 - Show how norbornance can be prepared from...Ch. 28 - Prob. 27PCh. 28 - Prob. 28PCh. 28 - Draw the product of each of the following...Ch. 28 - Prob. 30PCh. 28 - Prob. 31PCh. 28 - Prob. 32PCh. 28 - Prob. 33PCh. 28 - When the following compound is heated, a product...Ch. 28 - Prob. 35PCh. 28 - Propose a mechanism for the following reaction:Ch. 28 - Prob. 37PCh. 28 - Prob. 38PCh. 28 - Prob. 39PCh. 28 - Prob. 40PCh. 28 - If isomer A is heated to about 100 C, a mixture of...Ch. 28 - Propose a mechanism for the following reaction:Ch. 28 - Prob. 43PCh. 28 - A student found that heating any one of the...Ch. 28 - Prob. 45PCh. 28 - Prob. 46PCh. 28 - Prob. 47P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Differentiate between nuclear spin and electron spin.arrow_forwardDraw the trigonal prismatic MH6 molecular compound, where M is a 3d transition metal. a) Draw the trigonal prismatic MH6 molecular compound and determine its point group. b) i. What is the symmetry species for the 4s orbital on the central metal? ii. What is the symmetry species for the 3dx²-y² orbital on the central metal? Note: The z-axis is the principal axis. iii. Suggest a crystal field energy diagram for a d² electron configuration in a trigonal prismatic coordination environment. Label the metal d-orbital with their corresponding symmetry species label. Use the appropriate character table in the resource section.arrow_forwardPlease correct answer and don't used hand raiting don't used Ai solutionarrow_forward
- Don't used Ai solutionarrow_forwardShow work. Don't give Ai generated solutionarrow_forwardthis is an organic chemistry question please answer accordindly!! please post the solution draw the figures on a paper please hand drawn and post, please answer EACH part till the end and dont just provide wordy explanations, please draw them on a paper and post clearly!! answer the full question with all details EACH PART CLEARLY please thanks!! im reposting this please solve all parts and draw it not just word explanations!!arrow_forward
- A mixture of 0.412 M C12, 0.544 M F2, and 0.843 M CIF is enclosed in a vessel and heated to 2500 K. C12(g) + F2(g )2CIF(g) Kc = 20.0 at 2500 K Calculate the equilibrium concentration of each gas at 2500 K. [C12] = M [F2] = M [ CIF] =arrow_forwardShow reaction mechanism with explanation. don't give Ai generated solutionarrow_forwardShow work. Don't give Ai generated solutionarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Coenzymes and cofactors; Author: CH15 SWAYAM Prabha IIT Madras;https://www.youtube.com/watch?v=bubY2Nm7hVM;License: Standard YouTube License, CC-BY
Aromaticity and Huckel's Rule; Author: Professor Dave Explains;https://www.youtube.com/watch?v=7-BguH4_WBQ;License: Standard Youtube License