
(a)
Interpretation:
The given reaction has to be identified as electro cyclic reaction, a cycloaddition reaction or a sigma tropic reaction.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or broken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
There are mainly three types of pericyclic reactions,
- 1) Electrocyclic reactions
- 2) Cycloaddition reactions
- 3) Sigmatropic reactions
In an electrocyclic reaction “one new sigma- bond is formed or broken.”
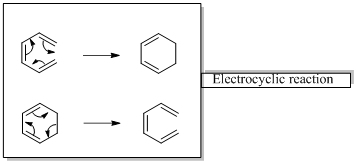
In a cycloaddition reaction “ two new sigma-bonds are formed or broken”
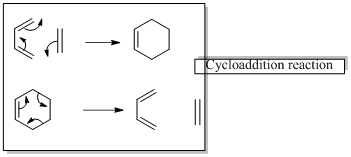
In a sigmatropic rearrangement reaction “ one new sigma-bond is formed as another breaks.”
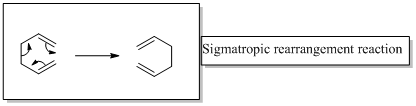
(b)
Interpretation: The given reaction has to be identified as electro cyclic reaction, a cycloaddition reaction or a sigma tropic reaction.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or broken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
There are mainly three types of pericyclic reactions,
- 1) Electrocyclic reactions
- 2) Cycloaddition reactions
- 3) Sigmatropic reactions
In an electrocyclic reaction “one new sigma- bond is formed or broken.”

In a cycloaddition reaction “ two new sigma-bonds are formed or broken”

In a sigmatropic rearrangement reaction “ one new sigma-bond is formed as another breaks.”

(c)
Interpretation: The given reaction has to be identified as electro cyclic reaction, a cycloaddition reaction or a sigma tropic reaction.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or broken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
There are mainly three types of pericyclic reactions,
- 1) Electrocyclic reactions
- 2) Cycloaddition reactions
- 3) Sigmatropic reactions
In an electrocyclic reaction “one new sigma- bond is formed or broken.”

In a cycloaddition reaction “ two new sigma-bonds are formed or broken”

In a sigmatropic rearrangement reaction “ one new sigma-bond is formed as another breaks.”

(d)
Interpretation: The given reaction has to be identified as electro cyclic reaction, a cycloaddition reaction or a sigma tropic reaction.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or broken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
There are mainly three types of pericyclic reactions,
- 1) Electrocyclic reactions
- 2) Cycloaddition reactions
- 3) Sigmatropic reactions
In an electrocyclic reaction “one new sigma- bond is formed or broken.”

In a cycloaddition reaction “ two new sigma-bonds are formed or broken”

In a sigmatropic rearrangement reaction “ one new sigma-bond is formed as another breaks.”

Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
Organic Chemistry (8th Edition)
- Draw the structure of compounds (E-H) from the following cycloaddition reactions.arrow_forwardDraw the structures of A - D from the following electrocyclic and cycloaddition reactions.arrow_forwardClassify each of the following cycloadditions and explain why orbital symmetry conservation rules allow or forbid each of the following reactions to occur as a concerted process.arrow_forward
- Show the structures for the major/significant organic product(s) expected for the following eleven reactions or sequences of reactions. If there is a major and minor productcircle the major product. Show all relevant stereochemistry.arrow_forwardDraw mechanism arrows and then predict the major products and stereochemistry. Also include the labels of thermodynamic and kinetic - no stick structure please dont understand like that. - 4- methypenta-1,3- diene with Hbrarrow_forwardRank the following alkenes in order of increasing stability (least to most stable)arrow_forward
- (d) Does the product have the same stereochemistry with the reactant? Explain. Instructions: Answer part (d) onlyarrow_forwardIdentify the mode of ring closure for each of the following electrocyclic reactions. a. Are the indicated hydrogens cis or transarrow_forwardDraw mechanism arrows and then predict the major products and stereochemistry. Also include the labels of thermodynamic and kinetic 3,4-dimethylhex-3-ene with HClarrow_forward
- Draw the mechanism and major product for each of the following Diels–Alder reactions. Pay attention to stereochemistry.Hint: You may need to consider conformations and orientations other than the ones shown.arrow_forwardfor the Cl2 addition reaction with 1,2dimethylcyclohexene, draw the most reasonable curved arrow mecanism. based on the reaction products, explain why a carbocation intermediate cannot be proposedarrow_forwardNeed all subparts. Should correct State which products are formed in the following reactions. Remember to include stereochemistry where relevant. In cases where no reaction takes place, write: "No reaction".arrow_forward
