
Concept explainers
Answer the following questions for this
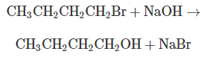
a. What is the rate expression for the reaction?
b. Draw the reaction profile for the reaction Label all parts Assume that the products are lower in energy than the reactants
c. What is the effect on the
d. What is the effect on the rate of the reaction of halving the concentration of sodium hydroxide?
Want to see the full answer?
Check out a sample textbook solution
Chapter 27 Solutions
General Chemistry: Principles and Modern Applications (11th Edition)
Additional Science Textbook Solutions
CHEMISTRY-TEXT
Chemistry (7th Edition)
Chemistry
Principles of Chemistry: A Molecular Approach (3rd Edition)
Principles of General, Organic, Biological Chemistry
Chemistry: Structure and Properties (2nd Edition)
- 3. Draw the products formed when each compound is treated with HNO3 and H2SO4. State whether the reaction occurs faster or slower than a similar reaction with be nzene. COCH3 CN OH .CI CH,CH3 a. b. C. d. e.arrow_forward2. Draw the products of each reaction. CH;CH2CI b. HNO3 Cl2 OCH3 Br -NO2 a. с. AICI3 H,SO4 FeCl3 3. Draw the products formed when each compound is treated with HNO3 and H2SO4. State whether the reaction occurs faster or slower than a similar reaction with be nzene.arrow_forwardWhich reagent(s) are used in the following reaction? 1. Hg(OAc), HO 2. NaBH₁ 1. BH 2. HO, OH Ch HO HO H₂SO4 NaNH, کی سسکے -OH and OHarrow_forward
- Q2. Classify the following reactions to: 1. Wagner- Meerwein rearrangement 2. Pinacolic Rearrangement 3. Beckmann Rearrangement 4. Baeyer-Villiger Rearrangement 5. Wolff Rearrangement H,SO4 1. OH OH Ag(I) 2. NO NO OH H,SO4 NH 3. OH H;C, H,SO, + H,O H 4. H;C CH, CH3 HO H,SO4 5. OH OH NH,OH H,SO4 NH 6. Aarrow_forwardIdentify the correct first mechanism step and final product of the reaction shown. 1. ВНз, THF 2. Н2О2, ОН" mechanism step final product H-BH2 OH А. H-BH2 H-BH2 OH C. H-BH2 LBH2 D. OH B.arrow_forwardThe following is a concerted, bimolecular reaction: CH3Br + NaCN → CH3CN + NaBr. a.What is the rate equation for this reaction? b.What happens to the rate of the reaction if [CH3Br] is doubled? c.What happens to the rate of the reaction if [NaCN] is halved? d.What happens to the rate of the reaction if [CH3Br] and [NaCN] are both increased by a factor of five?arrow_forward
- 1. What is the product of the following reaction? سد A. B. HO [H+] -H₂O (Η کھے D. HO Oarrow_forward1. Nucleophilic addition involving H, CO 2. Nucleophile elimination ?arrow_forward10 Predict which is the most reactive toward bromination of benzene? CH3 COOH CH3 COOH C. D. E. A B. CH3 COOH O A A O B B OCC OD D O E Earrow_forward
- 2. Draw the major product for each of the following reactions. a. a. Br Br 1.9-BBN 2. H₂O₂, NaOH 1. xs NaNH,/NH3 2. H₂Oarrow_forward1c. What is the nucleophile in the following reaction? .Br CH;CO0 Na LO OCCH3 NaBr ld. What is the electrophile in the following reaction? Br CH;COO Na L0OCCH3 NaBr +arrow_forwardThe following is a concerted, bimolecular reaction: CH3Br + NaCN ->CH3CN + NaBr.a. What is the rate equation for this reaction?b. What happens to the rate of the reaction if [CH3Br] is doubled?c. What happens to the rate of the reaction if [NaCN] is halved?d. What happens to the rate of the reaction if [CH3Br] and [NaCN] are both increased by a factor of five?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
