
Concept explainers
Substitution and Elimination Reactions
Answer the following questions for this E2 reaction:
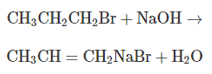 a. What is the rate expression for the reaction?
a. What is the rate expression for the reaction?
b. Draw the reaction profile for the reaction. Label all parts. Assume that the products are lower in energy than the reactants.
c. What is the effect on the
d. What is the effect on the rate of the reaction of halving the concentration of NaOH?
Want to see the full answer?
Check out a sample textbook solution
Chapter 27 Solutions
General Chemistry: Principles and Modern Applications (11th Edition)
Additional Science Textbook Solutions
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Introduction to Chemistry
Inorganic Chemistry
Organic Chemistry
Chemistry (7th Edition)
Organic Chemistry (8th Edition)
- 70. Determine the sole product of the following second order elimination reaction: NAOET, ETOH 400 K B D.arrow_forward7. In eight steps or less, convert the given starting materials into the desired products. eight steps or less a. b. Br CI eight steps or less HO, OHarrow_forwardWhat is the major product of the shown reaction ? CEC D2, Pd/C D D D D D -CEC -D A Deuterium (chemical symbol - D) is a heavy isotope of hydrogen. C O 1. C O2. B 3. D O 4. A this reaction won't happen as shown 5.arrow_forward
- Question 1 Consider the reaction below. The AGxn for this reaction is 100 kJ/mol: a. Sketch a free energy diagram of this reaction b. Indicate the AG*xn in your diagram c. Is this reaction endergonic or exergonic? Explain d. Draw the transition state for this reaction go +Harrow_forward1.What are the two products formed during the following reaction? F22+NH44I --> A.no reaction occurs B.NF2 and HI2 C.I2 and NH4F D.I2 and F2 E.I2F2 and NH4arrow_forwardWhat is the major product of the following reaction? A. B. NO₂ NO₂ NO₂ CH3CI, AICI3 C. D. NO₂ NO₂arrow_forward
- Consider the reaction H2S = S(s) + 2H* + 2e¯ This reaction can proceed via the addition of а. 13 b. I- O b aarrow_forwardConsider the following two-step reaction: a.How many bonds are broken and formed in Step [1]? Would you predict the ΔHo of Step [1] to be positive or negative? b.How many bonds are broken and formed in Step [2]? Would you predict the ΔHo of Step [2] to be positive or negative? c.Which step is rate-determining? d.Draw the structure for the transition state in both steps of the mechanism. e.If ΔHooverall is negative for this two-step reaction, draw an energy diagram illustrating all of the information in parts (a)–(d).arrow_forward4. What is the product of the following reaction? A. B. OH 1) CH3CH₂CH3MgBr 2) H₂O C. HO D.arrow_forward
- 5. w mexico What is the likely mechanism of this reaction? ew mexico Br A. E1cb B. E1 C. E2 D. None of the choices are correct. NaNH, DMF 6. I can answer question #5 because I know that: A. The solvent is polar protic, so a cation is easily formed B. The base is ionic, and so is a strong base C. The product follows Zaitsev's Rule D. The stability of the conjugate base allows for easy deprotonation as a first steparrow_forwardDraw an energy diagram for each reaction. Label the axes, the starting material, product, transition state, ?H°, and Ea.a. a concerted reaction with ?H° = –80 kJ/mol and Ea = 16 kJ/molb. a two-step reaction, A -> B ->C, in which the relative energy of the compounds is A < C < B, and the step A -> B is rate-determining.arrow_forward52. In a concerted or single step reaction, A. the rate of forward reaction is equal to the rate of backward reaction. B. the free energy of the reaction is negative C. the free energy of the reaction is positive D. the stability of the transition state controls the rate of the reaction.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
