
Concept explainers
(a)
Interpretation:
The hybridization of carbon atom, total C-C σ bonds and π bonds with 1â—¦,2â—¦,3â—¦ and 4â—¦ carbon atoms should be determined.
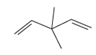
Concept introduction:
Hybridization is the process of mixing of atomic orbitals to form same energy and same shape hybrid orbitals which form covalent bond with overlapping of atomic orbitals of other atoms. The number of
1â—¦ orPrimary C atom = C atom which is bonded with one another C atom
2â—¦ orSecondary C atom= C atom which is bonded with two other C atoms
3â—¦ orTertiary C atom= C atom which is bonded with three other C atoms
4â—¦ or Quaternary C atom= C atom which is bonded with four other C atoms
(b)
Interpretation:
The hybridization of carbon atom, total C-C σ bonds and π bonds with 1â—¦, 2â—¦,3â—¦ and 4â—¦ carbon atoms should be determined.
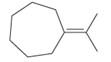
Concept introduction:
Hybridization is the process of mixing of atomic orbitals to form same energy and same shape hybrid orbitals which form covalent bond with overlapping of atomic orbitals of other atoms. The number of chemical bonds formed by the atom always determine type of hybridization. Sigma bonds could only form by hybrid orbitals whereas pi bonds are always formed by overlapping of un-hybrid orbitals hence hybridization is not part of pi bond formation.
1â—¦ or Primary C atom = C atom which is bonded with one another C atom
2â—¦ or Secondary C atom= C atom which is bonded with two other C atoms
3â—¦ or Tertiary C atom= C atom which is bonded with three other C atoms
4â—¦ or Quaternary C atom= C atom which is bonded with four other C atoms
(c)
Interpretation:
The hybridization of carbon atom, total C-C σ bonds and π bonds with 1â—¦, 2â—¦,3â—¦ and 4â—¦ carbon atoms should be determined.
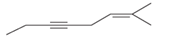
Concept introduction:
Hybridization is the process of mixing of atomic orbitals to form same energy and same shape hybrid orbitals which form covalent bond with overlapping of atomic orbitals of other atoms. The number of chemical bonds formed by the atom always determine type of hybridization. Sigma bonds could only form by hybrid orbitals whereas pi bonds are always formed by overlapping of un-hybrid orbitals hence hybridization is not part of pi bond formation.
1â—¦ or Primary C atom = C atom which is bonded with one another C atom
2â—¦ or Secondary C atom= C atom which is bonded with two other C atoms
3â—¦ or Tertiary C atom= C atom which is bonded with three other C atoms
4â—¦ or Quaternary C atom= C atom which is bonded with four other C atoms
Want to see the full answer?
Check out a sample textbook solution
Chapter 26 Solutions
General Chemistry: Principles and Modern Applications (11th Edition)
- Provide the IUPAC name for the following molecules. In your answer, include a description of how the name was derived. но Identify the hybridisation of all the carbon, oxygen and nitrogen atoms in the following molecules. OHarrow_forwardPredict the ideal values for the bond angles about each carbon atom in the following molecule. Indicate the hybridization of orbitals for each carbon. CH3CCCH2COOHarrow_forwardwhat is the molecular geometry of cyclopropane and the hybridization ?arrow_forward
- The remaining p orbital in an sp2 hybridized carbon contains electron. A pi-bond is formed by the overlap of p orbitals. Hence, in CH2=CH-CH=CH2.there are pi electrons.arrow_forwardIdentify the hybridization and bond angles at the carbon atoms in the molecule shown:arrow_forwardWhich of the following statements is untrue about the identity of the molecule?I. The molecule bond angles are associated with its sp-hybridized carbon.II. All the atoms are in a straight line where sigma & pi bonding exists.III. The molecule is acetylene having one sigma bond and 2 pi bonds.IV. A linear geometry of the molecule shows a highest bond strength and bond length compared to an alkane and alkene.arrow_forward
- The following compound is a bioactive natural product derived from marine sponges: a) Predict the orbital hybridization and geometry around each of the indicated atom. b) Name the functional groups present in the compound. c) Find the C-O bond(s) containing a C atom having a hybrid orbital with 50% s-character. Indicate this with an asterisk (*) in the structure. d) What is the molecular formula of the compound?arrow_forward7. The carbons indicated with the arrows in the following two compounds, show. OR OH A) The same type of hybridization on the carbon atom. B) The same geometry around the carbon atom. C) The same number of hydrogen atoms bonded to the carbon atom. D) Both carbons are involved in a bond. E) None of the above. 8. The molecular formula C₂H4O can be converted into three-line bond (Kekulé) structures that are consistent with valence rules. Which one of the following Kekulé structures is not consistent with valence rules that obey the octet rule? A) HO ||| HIC-CTH B) H C) / H D) H T 1 H H H H-C-C-H II HH H CU1O H 0-H O ANarrow_forwardMany reactions involve a change in hybridization of one or more atoms in the starting material. In following reaction, identify the atoms in the organic starting material that change hybridization and indicate the change.arrow_forward
- 5a) Construct a model of butene, C4H8, using two carbon atoms with planar geometry and two carbon atoms with tetrahedral geometry. How many other isomers of C₂H, can you construct? Draw a dash structural representation of each structure. 5b) How many bonds in butene enjoy free rotation?arrow_forwardSketch the hydrogen bond(s) that form between ethanol and nitrogen trihydride and dihydrogen monoxide. Show the proper VSEPR shapes of all molecules (meaning VSEPR shapes with proper bond angles) and use dotted or dashed lines to indicate the hydrogen bonds. Indicate 4 different H-bonds between: 1) ethanol and nitrogen trihydride 2) ethanol and dihydrogen monoxide 3) dihydrogen monoxide and nitrogen trihydride and then 4) between the two ethanol molecules.arrow_forwardConsider the skeletal structure in line‑angle (line‑bond) mode of 1‑isopropyl‑2‑methylcyclohexane. Identify the number of hydrogen atoms bound to each carbon in the structure.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning



