
Interpretation:
The percentages of neutral and protonated forms present in a solution of 0.0010M pyrimidine at pH = 7.3 are to be calculated if the pKa of pyrimidinium ion is 1.3.
Concept introduction:
pH And pKa are related by Henderson-Hasselbalch equation as
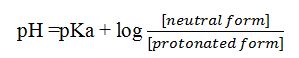
Knowing pH and pKa values, the ratio between the two forms and from which their percentages can be determined.
To calculate:
The percentages of neutral and protonated forms present in a solution of 0.0010M pyrimidine at pH = 7.3, if the pKa of pyrimidinium ion is 1.3.
Answer:
At pH = 7.3, almost 100% pyrimidine molecules exist in the neutral form.
Explanation:
Deprotonation of the ammonium ion of a base can be represented as,

The Henderson-Hasselbalch can be written as
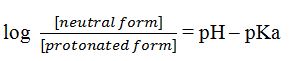
Substituting the values of pH and pKa, we get

Thus the concentration of neutral form is 106 times more than the protonated form. Hence almost 100% pyrimidine molecules exist in the neutral form.
Conclusion:
At pH = 7.3, almost 100% pyrimidine molecules exist in the neutral form.
Trending nowThis is a popular solution!

Chapter 24 Solutions
Organic Chemistry
- What are the concentrations of the various ionic species in a 0.1M solution of lysine at pH 4, 7, and 10?arrow_forwardThe ionization constant Ka for nitrous acid is 4.6x10⁻⁴. What are the the pKa for nitrous acid and the pH of a 0.1 M solution of this weak acid?arrow_forwardCalculate the pH of: A 0.0850 molar solution of the intermediate (HA) form of threonine. It is a polyprotic question.arrow_forward
- The pH of a 0.02M solution of a weak acid was measure at 4.6 a. What is the [H+] in this solution?b. Calculate the acid dissociation constant Ka and pKa for this acid?arrow_forwardNeutralization of phosphate . Given that phosphoricacid (H3PO4) can give up three protons with different pKa values, sketch a plot of pH as a function of added drops of sodium hydroxide solution, starting with a solution ofphosphoric acid at pH 1.0.arrow_forwardexplain anharmonicity.arrow_forward
- Calculate the pH of the solution after the addition of each of the given amounts of 0.0651 M HNO3 to a 50.0 mL solution of 0.0750 M aziridine. The pKa of aziridinium is 8.04. What is pH after adding 62.6 mL HNO3?arrow_forwardThe measured pH of a 0.100 M solution of triethylamine (NEt3) is 11.68. What pKa of triethylammonium (HNEt3+) is implied by this data? Enter your response to the nearest 0.01.arrow_forwardThe pKa values of the conjugate acids of 2-aminopyridine (6.71) and 4- aminopyridine (9.11) are as shown. Which nitrogen is the more basic in these structures and why?arrow_forward
- What is the pKb of the conjugate base of a weak acid that has a pKa equal to 3.52? (temperature is 25ºC)arrow_forwardCalculate the sodium acetate/acetic acid ratio to prepare a buffered solution with a pH of 5.06. (pKa = 4.76) 0.3:1 1.99:1 0.50:1 1.51:1 What is the correct answer? 1,2,3 or 4?arrow_forwardThe pKa values of the conjugate acids of 2-aminopyridine (6.71) and 4- aminopyridine (9.11) are as shown. Why are the acidities of the conjugate acids of these structures different by about two and a half orders of magnitude?arrow_forward
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning



