
Concept explainers
Give the IUPAC name for each of the following compounds:
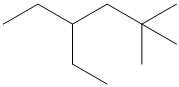
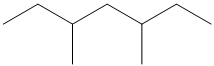
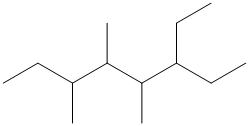
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Organic Chemistry - Standalone book
- Assign an IUPAC name to each of the following compounds. a. CH3(CH2)4SH b. CH3(CH2)4OHarrow_forwardArrange these compounds in order of increasing boiling point (values in C are 42, 24, 78, and 118). (a) CH3CH2OH (b) CH3OCH3 (c) CH3CH2CH3 (d) CH3COOHarrow_forwardGive the IUPAC name for following compound. CH3(CH2)3CH(CH2CH2CH3)CH(CH3)2arrow_forward
- Write the systematic (IUPAC) name for each of the following organic molecules: structure CH3CH2CH–CH2–CH–CH2CH–CH3 CH3 OH HO OH CH3 name 0 0 × Śarrow_forwardWhich of the following alcohols can NOT be oxidized? HO, CH2OH II III IV O IV III O1larrow_forwardWhat is the IUPAC name of the ether that has the structure shown? This molecule has the condensed formula C H 3 C H 2 C H 2 C H 2 O C H (C H 3) 2. IUPAC name:arrow_forward
- Give the IUPAC names of all the alcohols that have the formula C 5 H 12 Oarrow_forwardClassify each of the following hydrocarbons as saturated or unsaturated. a. CH3CH2CH=CH2 b. CH3CH3 c. CH2=CH2 d. CHCCH2CH3arrow_forwardWrite a condensed structural formula, such as CH3CH3, and describe the molecular geometry at each carbon atom. (a) propane. (b) 1-butanol. (c) ethyl propyl ether. (d) cis -4-bromo-2-heptene. (e) 2, 2, 3-trimethylhexane. (f) formaldehydearrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax





