
To review:
The chemical structures that are involved in the ionization of acetic acid in water.
Introduction:
The water is a polar and universal solvent. Most of the molecules having ionic bonds dissolve in water by making bonds with it. The compound dissociates into the individual ions making the ionic bond weaker than the solid form of the compound.
Explanation of Solution
The acetoacetic acid dissociates into the acetoacetate
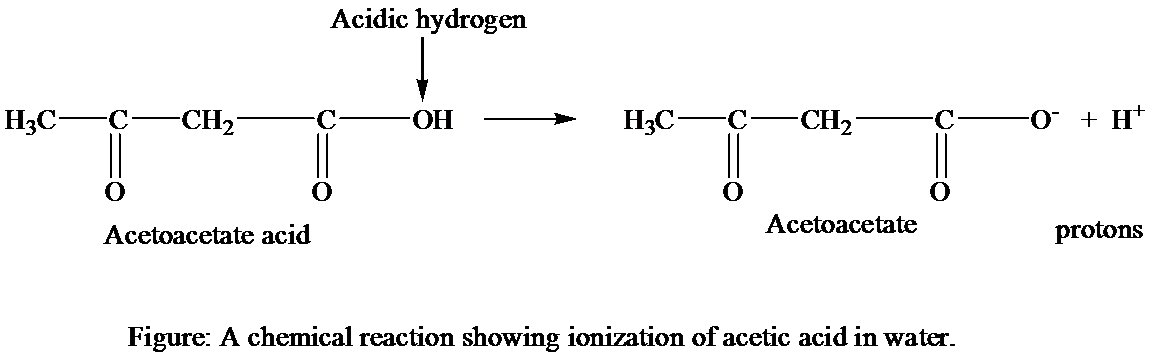
Thus, it can be concluded that the acetoacetate and hydrogen ions are chemical structures that are involved in the ionization of acetoacetic acid in water. The carboxyl group of acetoacetate infers the negative charge on the molecule and hydrogen ion gets dissociated from acetoacetic acid with its positive charge.
Want to see more full solutions like this?
Chapter 2 Solutions
Life: The Science of Biology
- Write the reaction, occurring in the liver, that causes theoxidation of ethanol. What is the product of this reactionand what symptoms are caused by the product?arrow_forwardDraw the structures of the following acids:(a) 2-Ethyl-3-hydroxyhexanoic acid (b) m-Nitrobenzoic acidarrow_forwardName the enantiomer of D-glucose.arrow_forward
- Write the complete reaction for the isomerization of citrate. Name the enzyme that catalyses these reaction andcprovide the formulas of the reactants and the product of these reaction ?arrow_forwardPropanamide and methyl acetate have about the same molar mass, both are quite soluble in water, and yet the boiling point of propanamide is 486 K, whereas that of methyl acetate is 330 K. Explain.arrow_forwardDraw a Fischer projection formula for the enantiomer of each of the following monosaccharides. (a to d)arrow_forward
- Which element is oxidized and which is reduced in the following reactions?arrow_forwardThe metabolic intermediate acetyl phosphate is an anhydride formed from acetic acid and phosphoric acid. What is the structure of acetyl phosphate?arrow_forwardIdentify the conjugate acid-base pairs in the following reactions: HNO2(aq) + H2O(l) → NO2 – (aq) + H3O+(aq) _______ ______ _________ ________ CH3NH2 + H2O(l) → CH3NH3+ + OH – _______ ________ ________ _________arrow_forward
- Draw the experimental setups for the preparation of benzoin and write the reaction mechanism.arrow_forwardIn the preparation of aspirin, You can do the functional group test to ensure the completion of the reaction. (a) What is the name of reagent used? (b) What is your observation if any unreacted starting material is present? (c) What is the name of the functional group responsible for this reaction?arrow_forwardWhat is the reverse of this reaction?arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





