
Interpretation:
The relation of size of the activation energy of an elementary step in a complex reaction and the rate of that step needs to be explained.
Concept introduction:
In a chemical reaction to be occurred; two most important requirements are activation energy to reactant molecule and correct orientation of reactant molecules to colloid and form product. The minimum energy that is required for the conversion reactant to product is called as activation energy.
Answer to Problem 38SSC
The high activation energy of an elementary step will slow down the rate of that step of the reaction.
Explanation of Solution
Given information:
The activation energy of an elementary step is related to the rate of that step.
A chemical reaction can involve one or multiple steps during the conversion of reactant to product.
Once the reactant molecules acquire activation energy, they form activated complex which is highly unstable due to highest energy and readily converts to product.
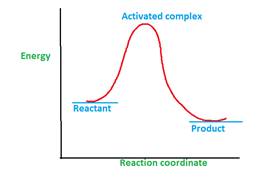
The elementary step with high activation energy will move with slow rate as it will take more time and energy for reactant molecules to acquire the activation energy and get convert to product. Thus high activation energy of an elementary step will slow down the rate of that step of the reaction.
The activation energy of an elementary step determines the rate of that step of the reaction.
Chapter 16 Solutions
Chemistry: Matter and Change
Additional Science Textbook Solutions
Chemistry: Structure and Properties (2nd Edition)
Chemistry: Structure and Properties
Introductory Chemistry (5th Edition) (Standalone Book)
Introductory Chemistry (6th Edition)
Organic Chemistry (9th Edition)
Essential Organic Chemistry (3rd Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





