
(a)
Interpretation:
Basic hydrolysis of triacylglycerol to form soap of below structure has to be determined.
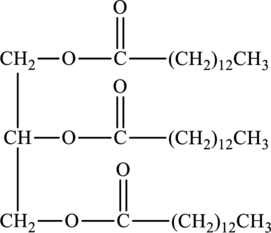
Concept Introduction:
Triacylglycerols are lipids that are formed by two or more similar or different fatty acids. They are also called triglycerides and are hydrophobic and nonpolar in nature. Addition of glycerol and three similar or different fatty acids results into triesters that is also triacylglycerols.
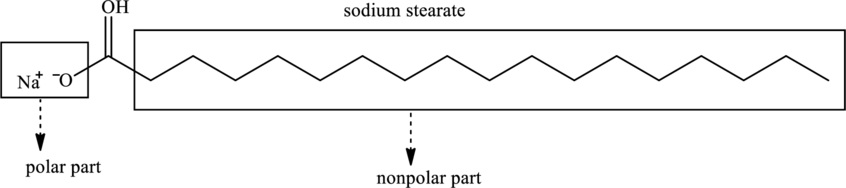
Soap is the long hydrocarbon chain with the metal salt of the fatty acid and is formed by the basic hydrolysis of triacylglycerol. For example, sodium stearate is the sodium salt of stearic acid as shown below.
Basic hydrolysis of triacylglycerol with water in presence of base produces metal salt of fatty acid and alcohol. The reaction that explains the basic hydrolysis of triacylglycerol is as follows:
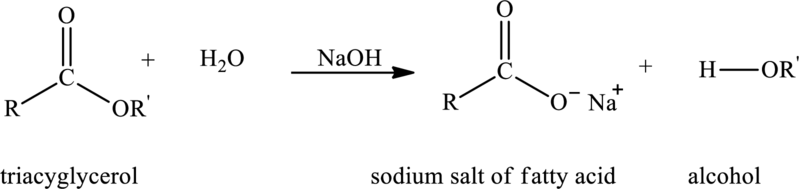
Here,
(b)
Interpretation:
Basic hydrolysis of triacylglycerol to form soap of below structure has to be determined.
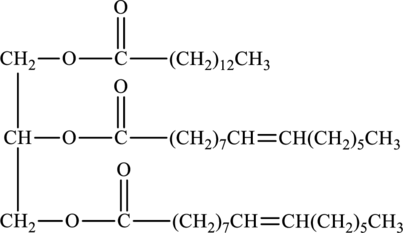
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Principles of General, Organic, Biological Chemistry
- Draw the general block diagram for a triacylglycerol.arrow_forwardDraw structures of glyceryl tripalmitate and glyceryl trioleate. Which would you expect to have a higher melting point?arrow_forwardThe plasmalogens are a group of lipids found in nerve and muscle cells. How do plasmalogens differ from fats?arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co





