
Concept explainers
(a)
Interpretation:
Product of hydrolysis of
Concept Introduction:
Lipids are
Hydrolysis of ester with water in presence of acid produces to give
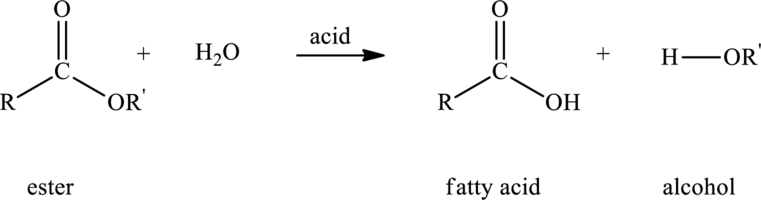
Here,
(b)
Interpretation:
Product of hydrolysis of
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Principles of General, Organic, Biological Chemistry
- Identify and label hemiacetal, acetal, hemiketal and ketal functional groups in the following molecules HO но. H. он он ÓH H. ÓH он но. CH2 .C H H. C OH ÓH но, CH2 OH OH HO, ÓHarrow_forwardWrite down the open reactions and products that occur with these reagents. | Compounds solubility in water. Benedict's Test lodoform Test Tollens' test H-COH CH;CH2OHarrow_forwardDetermine the product formed when butanone (CH3CH,COCH3) Is treated with each reagent. If no reaction occurs, label the reaction with "no reaction". PCC H2 Pd-C HO OH NazCr,O, H,SO.H,O [1] LIAIH, [2] H,O no reaction Ag20 NH,OH NABH, CH;OH Resetarrow_forward
- 2. Complete the following? Name the type of reaction? Draw the reactants and products? Name all the products unless its hemiacetal or acetal? If its acetal/ketal/hemiacetal/hemiketal identify the functional group? a. 2-Propanone H2 → Pd Propanal 2CH3OH – С. H+ d. Acetone Cu*2arrow_forwardtaken in order to gor the product Please explain the mechanisms/stepsarrow_forwardMacmillan Learning Name each compound. A. CH₂CH3 CH₂CH3arrow_forward
- 4. Number these in order of ease of hydrolysis. 1 = easiest to hydrolyze, 4 = hardest to hydrolyze. C.H.&CI C6H5CNHC(CH3)3 CH₂C=NCHCOCHarrow_forwardDraw the products formed when phenol(C6H5OH) is treated with each reagent. Give an explanation. d. (CH3CH2)2CHCOCl, AlCl3 j. product in (d), then NH2NH2, – OHarrow_forwardDraw the product formed when pentanal (CH3CH2CH2CH2CHO) is treated with each reagent. With some reagents, no reaction occurs.arrow_forward
- 3. Imitation vanilla extract contains vanillin and propylene glycol. Analysis via GC-MS shows a small quantity of a racemic mixture of a vanillin-propylene glycol acetal. Draw the structure of this acetal (considering stereochemistry) and briefly explain why it is racemic. HO H3CO vanillin H HO OH propylene glycolarrow_forwardDraw the product formed when pentanal (CH3 CH₂ CH₂ CH₂ CHO) is treated with each reagent. With some reagents, no reaction occurs. a. NaBH4, CH3OH b. [1] LiAiH4: [2] H₂O c. H₂, Pd-C d. PCC e. Na₂Cr₂O7, H₂SO4, H₂O f. Ag₂O, NH4OH g. [1] CH3 MgBr; [2] H₂O h. [1] C6H5 Li: [2] H₂O i. [1] (CH3)2 CuLi; [2] H₂O J. [1] HC=CNa; [2] H₂O k. [1] CH 3 C=CLI; [2] H₂O 1. The product in (a), then TBDMS-CI, imidazolearrow_forwardWrite down the reactions and products that occur with these reagents. Compounds solubility in water. Benedict's Test lodoform Test Jollens' test CH3-CH2-CHO CH2 3. CH- CH2-C-CH CH, H-COH CH;CH2OHarrow_forward
