
Concept explainers
(a)
Interpretation:
Rings of the cholesterol steroid have to be labeled.
Concept Introduction:
Group of lipids that consist of 1 five-membered ring and 3 six-membered rings in its skeleton structure are called steroids. Steroids differ with each other in terms of the attachment of the substituent and thus this changes their identity. The skeleton structure of steroid is as follows:
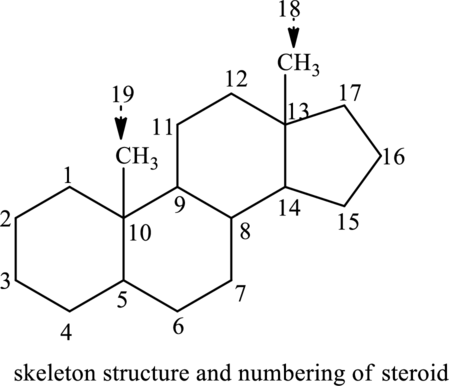
(b)
Interpretation:
Carbon number to that
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
Location of double bond in cholesterol has to be determined.
Concept Introduction:
Refer to part (a).
(d)
Interpretation:
Polar bonds in cholesterol and insolubility of cholesterol in water have to be explained.
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Principles of General, Organic, Biological Chemistry
- Draw the structure of a glycosphingolipid molecule with an N-acetyl glucosamine (B-1 linkage) head group and a linoleic acid attached. Number each of the carbons in linoleic acid.arrow_forwardExplain how α-tocopherol and ascorbate work together in suppressing the oxidation of polyunsaturated fatty acids (PUFAs).arrow_forwardWhat molecular interactions explain the observed solubility class of urea?arrow_forward
- What are the sterioisomers of mannuronic acid?arrow_forward(Chemical Connections 30A) What is a recommendation from ChooseMyPlate.gov regarding dairy products?arrow_forwardExplain why insulin (a peptide hormone that regulates carbohydrate metabolism) is administered by injection rather than in oral dosage form.arrow_forward
- 1. Describe the difference between collagen, alpha keratin and beta keratin under the following headings: 1.1. Amino acid composition 1.2. Carbon dioxide poisoning 1.3. Three dimensional structure 1.4. Where is it foundarrow_forwardWhich among the statements below explain(s) why fructose gives a positive result to the Tollen’s test? A. Fructose decomposes into an aldehyde under basic conditions. B. Fructose isomerizes to glucose in aqueous ammonia. C. Fructose is reduced to mannitol. D. Ap+ ions oxidize ketones to carboxylic acids. Kindly explain your answer in detail.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,



