
Concept explainers
(a)
Interpretation:
Product of hydrolysis of below triacylglycerol in presence of water and sulfuric acid has to be determined.
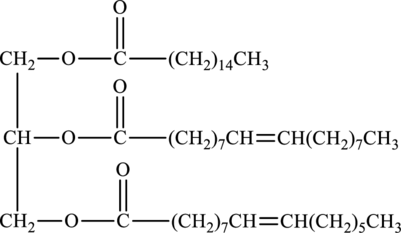
Concept Introduction:
Hydrolysis of ester with water in presence of acid produces
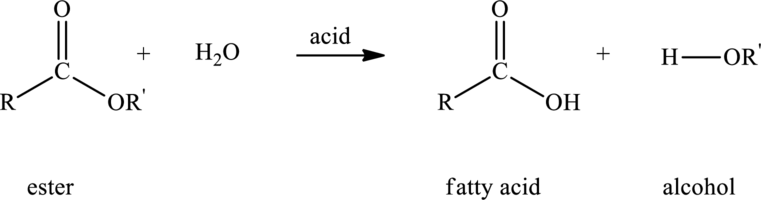
Here,
(b)
Interpretation:
Product of hydrolysis of triacylglycerol in presence of water and sodium hydroxide has to be determined.
Concept Introduction:
Basic hydrolysis of triacylglycerol with water in presence of base produces metal salt of fatty acid and alcohol. The reaction that explains the basic hydrolysis of triacylglycerol is as follows:
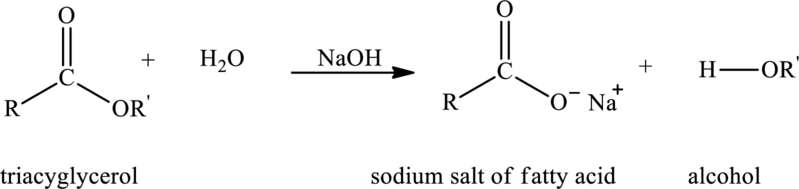
Here,
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Principles of General, Organic, Biological Chemistry
- Draw condensed structural formulas for all products you would obtain from the saponification with KOH of the triacylglycerol in Problem 19-54.arrow_forwardWhat products are formed when benzoic acid (C 6H 5COOH) is treated with each base: (a) NaOH; (b) Na 2CO 3; (c) NaHCO 3?arrow_forwardIdentify the polar and nonpolar regions in the following fatty acid: но Is the fatty acid saturated or unsaturated? Is the fatty acid an omega 3, omega 6 or neither?arrow_forward
- Draw condensed structural formulas for all products obtained from the complete hydrolysis of the following triacylglycerol under acidic conditions.arrow_forwardWhich of the following is an unsaturated triacylglycerol? 요 0 FO 0- =ㅁ 아 -NH5arrow_forwardPlease don't provide handwritten solutionarrow_forward
- 1. Which of the following are saponifiable lipids? a) triacylglycerols; f) stepoids3; b) prostaglandins3B g) glycolipids; c) waxes;B h) terpenes; d) leukotrienes; i) phosphoglycerides; e) sphyngolipids; k) plasmalogens?arrow_forwardConsider the structures of the two fatty acids shown below.arrow_forwardTriacylglycerides obtained from animal fats and plant oils are typically used in the production of biodiesel. Explain the advantage of using a triacylglyceride as the starting material for biodiesel synthesis over the waxy esters found in Jojoba oil.arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning



