
Concept explainers
(a)
Interpretation: The Lewis structure for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
Formal charge on each atom can be determined with the help of number of valence shell electrons, number of lone pair electrons and bond pair electrons. The formula for the formal charge can be written as:
(a)
Answer to Problem 85E
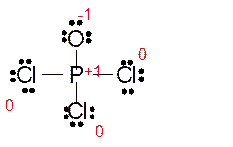
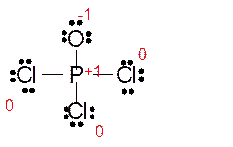
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
(b)
Interpretation: The Lewis structure for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
Formal charge on each atom can be determined with the help of number of valence shell electrons, number of lone pair electrons and bond pair electrons. The formula for the formal charge can be written as:
(b)
Answer to Problem 85E
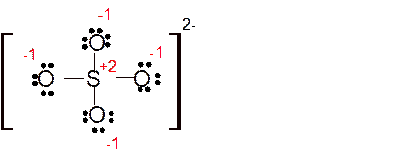
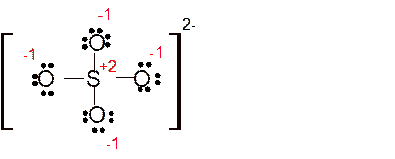
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
(c)
Interpretation: The Lewis structure for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
Formal charge on each atom can be determined with the help of number of valence shell electrons, number of lone pair electrons and bond pair electrons. The formula for the formal charge can be written as:
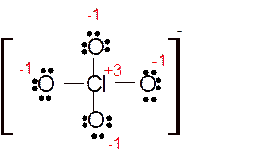
(c)
Answer to Problem 85E
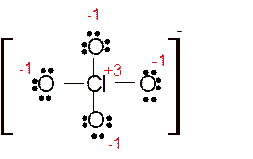
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
(d)
Interpretation: The Lewis structure for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
Formal charge on each atom can be determined with the help of number of valence shell electrons, number of lone pair electrons and bond pair electrons. The formula for the formal charge can be written as:
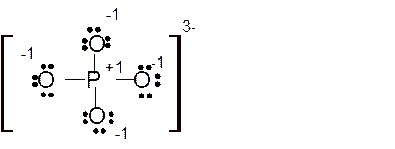
(d)
Answer to Problem 85E
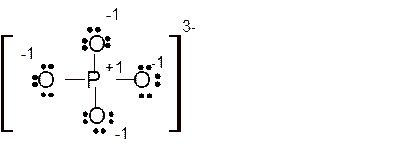
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
(e)
Interpretation: The Lewis structure for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
Formal charge on each atom can be determined with the help of number of valence shell electrons, number of lone pair electrons and bond pair electrons. The formula for the formal charge can be written as:
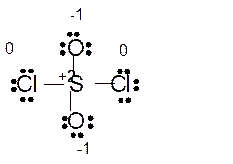
(e)
Answer to Problem 85E
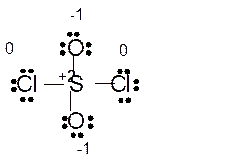
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
(f)
Interpretation: The Lewis structure for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
Formal charge on each atom can be determined with the help of number of valence shell electrons, number of lone pair electrons and bond pair electrons. The formula for the formal charge can be written as:
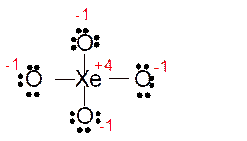
(f)
Answer to Problem 85E
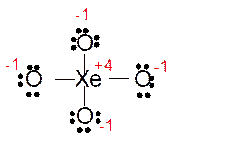
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
(g)
Interpretation: The Lewis structure for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
Formal charge on each atom can be determined with the help of number of valence shell electrons, number of lone pair electrons and bond pair electrons. The formula for the formal charge can be written as:
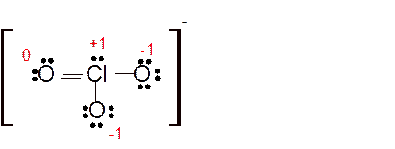
(g)
Answer to Problem 85E
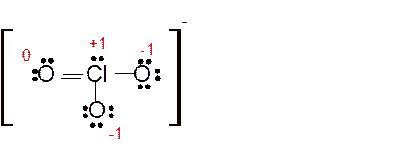
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
(h)
Interpretation: The Lewis structure for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
Formal charge on each atom can be determined with the help of number of valence shell electrons, number of lone pair electrons and bond pair electrons. The formula for the formal charge can be written as:
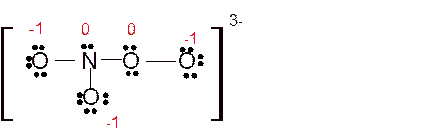
(h)
Answer to Problem 85E
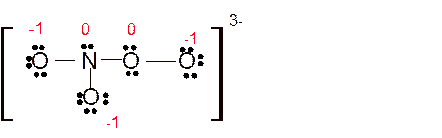
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
Want to see more full solutions like this?
Chapter 13 Solutions
Chemical Principles
- What is meant by a chemical bond? Why do atoms form bonds with each other? Why do some elements exist as molecules in nature instead of as free atoms?arrow_forwardDefine formal charge and explain how to calculate it. What is the purpose of the formal charge? Organic compounds are composed mostly of carbon and hydrogen but also may have oxygen, nitrogen, and/or halogens in the formula. Formal charge arguments work very well for organic compounds when drawing the best Lewis structure. How do C, H, N, O, and Cl satisfy the octet rule in organic compounds so as to have a formula charge of zero?arrow_forwardThese are NOTlegitimate Lewisstructures (and aremissing formalcharges). Show (as inthe example) how apair of electrons canbe moved to make theLewis structurelegitimate.arrow_forward
- Bond Enthalpy When atoms of the hypothetical element X are placed together, they rapidly undergo reaction to form the X2 molecule: X(g)+X(g)X2(g) a Would you predict that this reaction is exothermic or endothermic? Explain. b Is the bond enthalpy of X2 a positive or a negative quantity? Why? c Suppose H for the reaction is 500 kJ/mol. Estimate the bond enthalpy of the X2 molecule. d Another hypothetical molecular compound, Y2(g), has a bond enthalpy of 750 kJ/mol, and the molecular compound XY(g) has a bond enthalpy of 1500 kJ/mol. Using bond enthalpy information, calculate H for the following reaction. X2(g)+Y2(g)2XY(g) e Given the following information, as well as the information previously presented, predict whether or not the hypothetical ionic compound AX is likely to form. In this compound, A forms the A+ cation, and X forms the X anion. Be sure to justify your answer. Reaction: A(g)+12X2(g)AX(s)The first ionization energy of A(g) is 400 kJ/mol. The electron affinity of X(g) is 525 kJ/mol. The lattice energy of AX(s) is 100 kJ/mol. f If you predicted that no ionic compound would form from the reaction in Part e, what minimum amount of AX(s) lattice energy might lead to compound formation?arrow_forwardPredict die molecular structure and bond angles for each molecule or ion in Exercises 88 and 94. a. POCl3, SO42, XeO4, PO43, ClO4 b. NF3, SO32, PO33, ClO3 c.ClO2, SCl2, PCl2 d. Considering your answers to parts a, b, and c. what conclusions can you draw concerning the structures of species containing the same number of atoms and the same number of valence electrons? (O3), sulfur dioxide, and sulfur trioxide.arrow_forwardWhich of the species with octets in Question 31 are dipoles?arrow_forward
- Write Lewis structures for the following: (a) H2 (b) HBr (c) PCl3 (d) SF2 (e) H2CCH2 (f) HNNH (g) H2CNH (h) NO (i) N2 (j) CO (k) CNarrow_forward18.What are the formal charges present in in the ion CH3NH3+? Assume all atoms other an hydrogen have an octet and that there are two central atoms. A. N is +1 and C is 0 B. N is +1 and C is -1 C. N is +2 and C is -1 D. N is 0 and C is 0 E. N is 0 and C is +1arrow_forwardWrite Lewis structures for each molecule or ion. Include reso- nance structures if necessary and assign formal charges to all atoms. If necessary, expand the octet on the central atom to lower formal charge. a. SO,2- b. HSO, c. SO3 d. BrOzarrow_forward
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning





