
Concept explainers
(a)
Interpretation: The Lewis structure for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that existing on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
Formal charge on each atom can be determined with the help of number of valence shell electrons, number of lone pair electrons and bond pair electrons. The formula for the formal charge can be written as:
(a)
Answer to Problem 86E
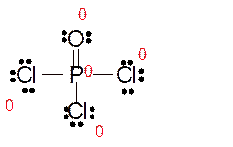
Explanation of Solution
Total number of valence electrons in
Hence the best Lewis structure for
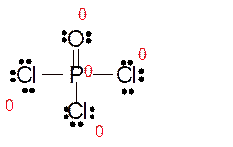
Here all bonded atoms have minimum formal charge therefore it must be the best Lewis structure.
(b)
Interpretation: The Lewis structure for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that existing on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
Formal charge on each atom can be determined with the help of number of valence shell electrons, number of lone pair electrons and bond pair electrons. The formula for the formal charge can be written as:
(b)
Answer to Problem 86E
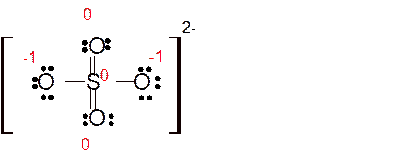
Explanation of Solution
Total number of valence electrons in
Hence the best Lewis structure for
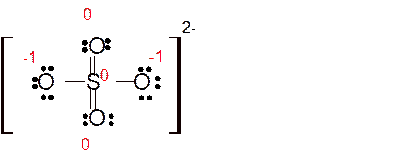
Here all bonded atoms have minimum formal charge therefore it must be the best Lewis structure.
(c)
Interpretation: The Lewis structure for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that existing on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
Formal charge on each atom can be determined with the help of number of valence shell electrons, number of lone pair electrons and bond pair electrons. The formula for the formal charge can be written as:
(c)
Answer to Problem 86E
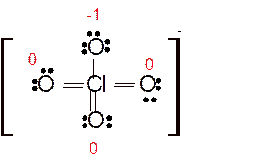
Explanation of Solution
Total number of valence electrons in
Hence the best Lewis structure for
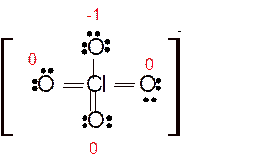
Here all bonded atoms have minimum formal charge therefore it must be the best Lewis structure.
(d)
Interpretation: The Lewis structure for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that existing on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
Formal charge on each atom can be determined with the help of number of valence shell electrons, number of lone pair electrons and bond pair electrons. The formula for the formal charge can be written as:
(d)
Answer to Problem 86E
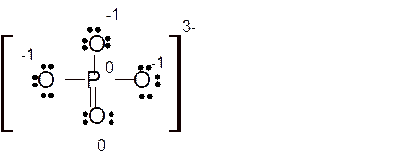
Explanation of Solution
Total number of valence electrons in
Hence the best Lewis structure for
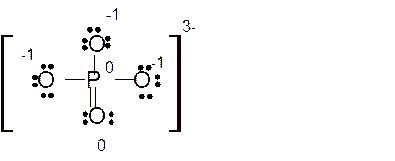
Here all bonded atoms have minimum formal charge therefore it must be the best Lewis structure.
(e)
Interpretation: The Lewis structure for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that existing on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
Formal charge on each atom can be determined with the help of number of valence shell electrons, number of lone pair electrons and bond pair electrons. The formula for the formal charge can be written as:
(e)
Answer to Problem 86E
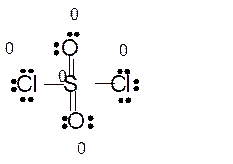
Explanation of Solution
Total number of valence electrons in
Hence the best Lewis structure for
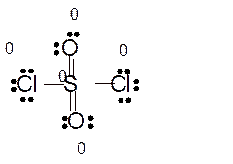
Here all bonded atoms have minimum formal charge therefore it must be the best Lewis structure.
(f)
Interpretation: The Lewis structure for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that existing on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
Formal charge on each atom can be determined with the help of number of valence shell electrons, number of lone pair electrons and bond pair electrons. The formula for the formal charge can be written as:
(f)
Answer to Problem 86E
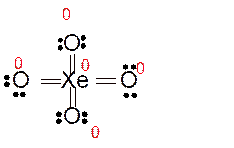
Explanation of Solution
Total number of valence electrons in
Hence the best Lewis structure for
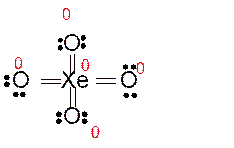
Here all bonded atoms have minimum formal charge therefore it must be the best Lewis structure.
(g)
Interpretation: The Lewis structure for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that existing on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
Formal charge on each atom can be determined with the help of number of valence shell electrons, number of lone pair electrons and bond pair electrons. The formula for the formal charge can be written as:
(g)
Answer to Problem 86E
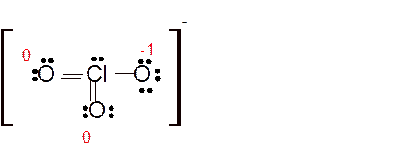
Explanation of Solution
Total number of valence electrons in
Hence the best Lewis structure for
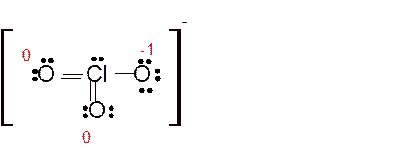
Here all bonded atoms have minimum formal charge therefore it must be the best Lewis structure.
(h)
Interpretation: The Lewis structure for
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that existing on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
Formal charge on each atom can be determined with the help of number of valence shell electrons, number of lone pair electrons and bond pair electrons. The formula for the formal charge can be written as:
(h)
Answer to Problem 86E
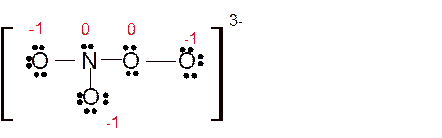
Explanation of Solution
Total number of valence electrons in
Hence the best Lewis structure for
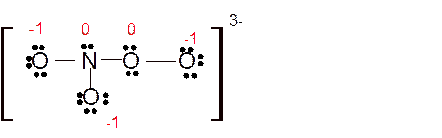
Here all bonded atoms have minimum formal charge therefore it must be the best Lewis structure.
Want to see more full solutions like this?
Chapter 13 Solutions
Chemical Principles
- Represent the bonding in SF2 (F-S-F) with Lewis diagrams. Include the formal charges on all atoms. The dimerof this compound has the formula S2F4. It was isolated in1980 and shown to have the structure F3S-SF. Draw apossible Lewis diagram to represent the bonding in thedimer, indicating the formal charges on all atoms. Is it possible to draw a Lewis diagram for S2F4 in which all atomshave valence octets? Explain why or why notarrow_forwardLewis structures for CNS_include formal charges on atoms with non-zero values. Concisel identify at least four steps of the procedure for drawing the Lewis structures (3d structure with stating geometry)arrow_forwardThe same student concluded by saying “Bond type can be calculated by subtracting the electronegativities of the elements involved in a chemical bond. Electronegativity differences below 1.7 are covalent bonds, and electronegativity differences 0.4 and below are non-polar covalent bonds and between 0.4 to 1.6 are considered by most to be polar covalent bonds.” Do you agree with this student? Justify your response.arrow_forward
- For the structure of IF 5 shown below: f ** The formal charge of the I would be The formal charge of each F would be A A For the structure of IF 5 shown below: The formal charge of the I would be The formal charge of each F would be A/arrow_forward1. For the species listed above, write two valid and likely Lewis structures. 2. Calculate formal charges on all atoms in each structure. Apply the criteria mentioned above to select the “best” Lewis structure. Label your chosen Lewis structure for each species as “BEST”.arrow_forwardA stable triatomic molecule can be formed that contains one atom each of nitrogen, sulfur, and fluorine. Three bonding structures are possible, depending on which is the central atom: NSF, SNF, and SFN. (a) Write a Lewis diagram for each of these molecules, indicating the formal charge on each atom. (b) Often, the structure with the least separation of formal charge is the most stable. Is this statement consistent with the observed structure for this molecule—namely, NSF, which has a central sulfur atom? (c) Does consideration of the electronegativities of N, S, and F from Figure 3.18 help rationalize this observed structure? Explain.arrow_forward
- Derive Lewis structures for the compounds below. Furanarrow_forwardTwo possible Lewis diagrams for sulfine (H2CSO) are (a) Compute the formal charges on all atoms. (b) Draw a Lewis diagram for which all the atoms in sulfine have formal charges of zero.arrow_forwardThe skeleton of acetyl chloride is . Write the best Lewis structure for acetyl chloride by following the procedure in the previous problem.arrow_forward
- An incomplete structure of a porphyrin ring is shown below. The structure is missing three pi bonds and does not show the non-zero formal charges on two atoms. Complete the structure of the porphyrin ring with the missing pi bonds and include all of the formal charges..arrow_forwardUse three rules to evaluate the relative energies of two or more validresonance structures ?arrow_forwardDetermine the formal charge for the indicated atoms in the following species or compounds below.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,




