
Concept explainers
(a)
Interpretation: The species which have same number of atoms and same number of valence electrons in
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The sum of valence electrons must be arranged in such a way that all atoms must get octet configuration (8 electrons).
(a)
Answer to Problem 65E
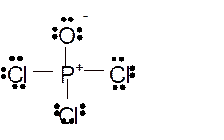
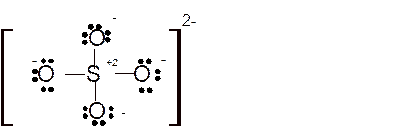
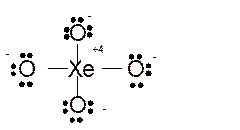
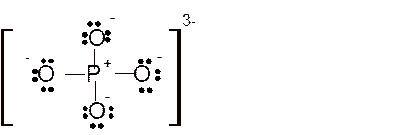
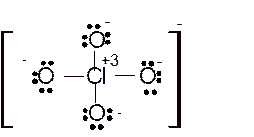
Hence here all
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
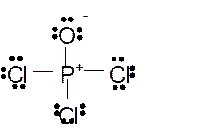
Total number of valence electrons in
Hence the best Lewis structure for
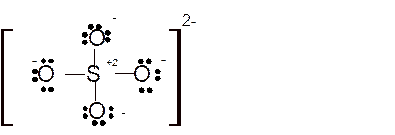
Total number of valence electrons in
Hence the best Lewis structure for
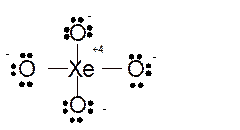
Total number of valence electrons in
Hence the best Lewis structure for
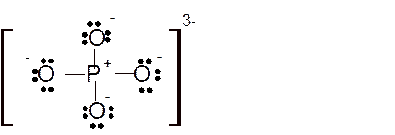
Total number of valence electrons in
Hence the best Lewis structure for
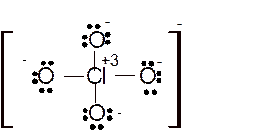
Hence here all
(b)
Interpretation: The species which have same number of atoms and same number of valence electrons in
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The sum of valence electrons must be arranged in such a way that all atoms must get octet configuration (8 electrons).
(b)
Answer to Problem 65E
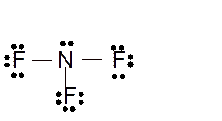
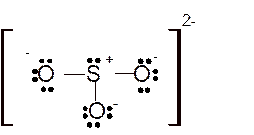
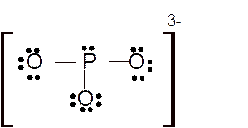
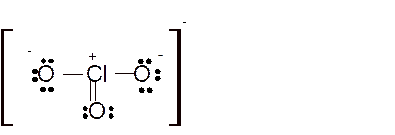
Hence here all
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
Total number of valence electrons in
Hence the best Lewis structure for
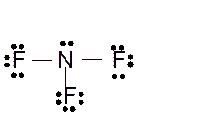
Total number of valence electrons in
Hence the best Lewis structure for
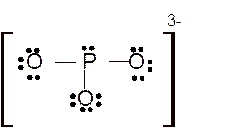
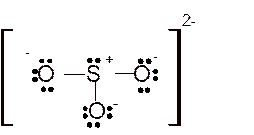
Total number of valence electrons in
Hence the best Lewis structure for
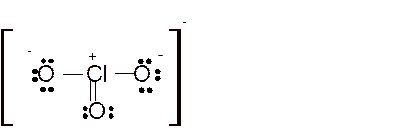
Hence here all
(c)
Interpretation: The species which have same number of atoms and same number of valence electrons in
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that exist on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The sum of valence electrons must be arranged in such a way that all atoms must get octet configuration (8 electrons).
(c)
Answer to Problem 65E
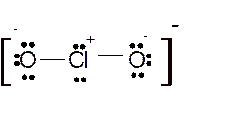
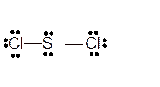
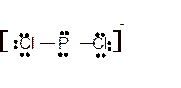
Hence here all
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
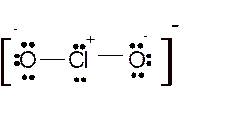
Total number of valence electrons in
Hence the best Lewis structure for
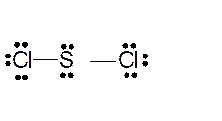
Total number of valence electrons in
Hence the best Lewis structure for
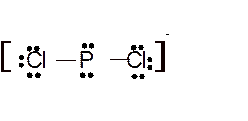
Hence here all
Want to see more full solutions like this?
Chapter 13 Solutions
Chemical Principles
- Give detailed Solution with explanation neededarrow_forwardRank the following in increasing order of nonmetallic character by numbering them in order from 1 through 6, with 1 being the least nonmetallic and 6 being the most nonmetallic. Se Cr K Br Са Asarrow_forwardThree biological ly important diatomic species. eitherbecause they promote or inhibit life. are (a) CO. (b) NO. and(c) CN-. The first binds to haemoglobin. the second is a neurotransmitter. and the third interrupts the respiratory electrontransfer chain. Their biochemical action is a reflection of theirorbital structure. Deduce their ground-state electron configurations. For heteronuclear diatomic molecules. a good firstapproximation is that the energy level diagram is much thesame as for homonuclear diatomic moleculesarrow_forward
- The following sketches show the atomic orbital wave functions(with phases) used to construct some of the MOs ofa homonuclear diatomic molecule. For each sketch, determinethe type of MO that will result from mixing theatomic orbital wave functions as drawn. Use the same labelsfor the MOs as in the “Closer Look” box on phases.arrow_forwardGive detailed Solution with explanation needed..don't give Handwritten answerarrow_forwardPlease answer both parts n detailarrow_forward
- The Be2 molecule has been detected experimentally. It has a bond length of 2.45 Å and a bond dissociation energy of 9.46kJmol1 . Write the ground-state electron configuration of Be2 and predict its bond order using the theory developed in the text. Compare the experimental bonding data on Be2 with those recorded for B2,C2,N2, and O2 in Table 6.3. Is the prediction that stems from the simple theory significantly incorrect?arrow_forwardCalcium carbide (CaC2) is an intermediate in the manufacturing of acetylene (C2H2) . It is the calcium salt of the carbide (also called acetylide) ion (C22) . What is the electron configuration of this molecular ion? What is its bond order?arrow_forwardFollowing the pattern of Figure 6.21, work out the correlation diagram for the BeN molecule, showing the relative energy levels of the atoms and the bonding and antibonding orbitals of the molecule. Indicate the occupation of the MOs with arrows. State the order of the bond and comment on the magnetic properties of BeN.arrow_forward
- In a gaseous RbF molecule, the bond length is 2.2741010m . Using data from Appendix F and making the same oversimplified assumption as in the prior problem on the shape of the potential curve from Rb++F to an internuclear separation of 2.2741010m , calculate the energy in kJmol1 required to dissociate RbF to neutral atoms.arrow_forwardThe photoelectron spectrum of HBr has two main groups of peaks. The first has ionization energy 11.88 eV. The next peak has ionization energy 15.2 eV, and it is followed by a long progression of peaks with higher ionization energies. Identify the molecular orbitals corresponding to these two groups of peaks.arrow_forwardCompare the electron density in the 1g and 1u* molecular orbitals for H2+ shown in Figure 6.5 with the classical model for bonding for H2+ summarized in Figure 3.22b. Which of these molecular orbitals describes the bond in H2+ ?arrow_forward

 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,





