
Concept explainers
(a)
Interpretation: The value of
Concept Introduction:
A chemical compound can be formed by either ionic bond or covalent bond between bonded atoms. The ionic compound is formed by opposite charge ions; cations and anions. The covalent compound is formed by sharing of electrons between bonded atoms.
The bond energy of a chemical bond can be defined as the energy required to break that chemical bond. The bond energy that is needed to break the bonds in reactant molecule and the energy released to form
(a)
Answer to Problem 48E
Explanation of Solution
Given:
For the given reaction
Calculate
Hence
(b)
Interpretation: The value of
Concept Introduction:
A chemical compound can be formed by either ionic bond or covalent bond between bonded atoms. The ionic compound is formed by opposite charge ions; cations and anions. The covalent compound is formed by sharing of electrons between bonded atoms.
The bond energy of a chemical bond can be defined as the energy required to break that chemical bond. The bond energy that is needed to break the bonds in reactant molecule and the energy released to form chemical bonds in product can be used to calculate the
(b)
Answer to Problem 48E
Explanation of Solution
Given:
For the given reaction from the bond energy
Calculate
Hence,
(c)
Interpretation: The value of
Concept Introduction:
A chemical compound can be formed by either ionic bond or covalent bond between bonded atoms. The ionic compound is formed by opposite charge ions; cations and anions. The covalent compound is formed by sharing of electrons between bonded atoms.
The bond energy of a chemical bond can be defined as the energy required to break that chemical bond. The bond energy that is needed to break the bonds in reactant molecule and the energy released to form chemical bonds in product can be used to calculate the
(c)
Answer to Problem 48E
Hence
Explanation of Solution
Given:
For the given reaction
Calculate
Hence
(d)
Interpretation: The value of
for the given reaction in gas phase needs to be determined with the help of bond energy value:
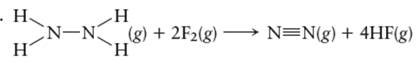
Concept Introduction:
A chemical compound can be formed by either ionic bond or covalent bond between bonded atoms. The ionic compound is formed by opposite charge ions; cations and anions. The covalent compound is formed by sharing of electrons between bonded atoms.
The bond energy of a chemical bond can be defined as the energy required to break that chemical bond. The bond energy that is needed to break the bonds in reactant molecule and the energy released to form chemical bonds in product can be used to calculate the
(d)
Answer to Problem 48E
Explanation of Solution
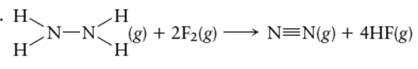
For the given reaction
Calculate
Hence
Want to see more full solutions like this?
Chapter 13 Solutions
Chemical Principles
- The bond energy for a CH bond is about 413 kJ/mol in CH4 but 380 kJ/mol in CHBr3. Although these values are relatively close in magnitude, they are different. Explain why they are different. Does the fact that the bond energy is lower in CHBr3, make any sense? Why?arrow_forwardThe standard enthalpies of formation for S(g), F(g), SF4(g), and SF6(g) are +278.8, +79.0, 775, and +1209 KJ/mol, respectively. a. Use these data to estimate the energy of an SF bond. b. Compare your calculated value to the value given in Table 3-3. What conclusions can you draw? c. Why are the Hf values for S(g) and F(g) not equal to zero, since sulfur and fluorine are elements?arrow_forwardCompare your answers from parts a and b of Exercise 69 of Chapter 3 with H values calculated for each reaction using standard enthalpies of formation in Appendix 4. Do enthalpy changes calculated from bond energies give a reasonable estimate of the actual values?arrow_forward
- Estimate H for the following reactions using bond energies given in Table 8.5. 3CH2=CH2(g) + 3H2(g) 3CH2CH3(g) The enthalpies of formation for C6H6(g) and C6H12 (g) are 82.9 and 90.3 kJ/mol. respectively. Calculate H for the two reactions using standard enthalpies of formation from Appendix 4. Account for any differences between the results obtained from the two methods.arrow_forwardCompare your answers from parts a and b of Exercise 69 with H values calculated for each reaction using standard enthalpies of formation in Appendix 4. Do enthalpy changes calculated from bond energies give a reasonable estimate of the actual values?arrow_forwardThe reaction of quicklime, CaO, with water produces slaked lime, Ca(OH)2, which is widely used in the construction industry to make mortar and plaster. The reaction of quicklime and water is highly exothermic: CaO(s)+H2O(l)Ca(OH)2(s)H=350kJmol1 (a) What is the enthalpy of reaction per gram of quicklime that reacts?. (b) How much heat, in kilojoules, is associated with the production of 1 ton of slaked lime?arrow_forward
- Given the following data: NO2(g) NO(g) + O(g)H = 233 kJ 2O3(g) 3O2(g)H = 427 kJ NO(g) + O3(g) NO2(g) + O2(g)H = 199 kJ Calculate the bond energy for the O2 bond, that is, calculate H for: O2(g) 2O(g)H = ?arrow_forwardCalculate the enthalpy of reaction for HCN(g)H(g)+C(g)+N(g) from enthalpies of formation (see Appendix C). Given that the CH bond enthalpy is 411 kJ/mol, obtain a value for the CN bond enthalpy. Compare your result with the value given in Table 9.5.arrow_forwardExplain the decomposition of nitroglycerin in terms of relative bond enthalpies.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





