
Concept explainers
(a)
Interpretation:
The structural formula of the ester formed by reaction between formic acid and methanol has to be drawn.
Concept Introduction:
Esters are a class of compound that is formed by the reaction between an alcohol and a 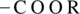 . Hydrolysis of ester gives back an alcohol and a carboxylic acid.
. Hydrolysis of ester gives back an alcohol and a carboxylic acid.
(b)
Interpretation:
The structural formula of the ester formed by reaction between butyric acid and ethanol has to be drawn.
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The structural formula of the ester formed by reaction between acetic acid and 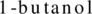 has to be drawn.
has to be drawn.
Concept Introduction:
Refer to part (a).
(d)
Interpretation:
The structural formula of the ester formed by reaction between propanoic acid and 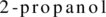 has to be drawn.
has to be drawn.
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
Chemistry: The Molecular Science
- Identify which of the following statement(s) is/are true. (f) Aldehydes, ketones, carboxylic acids, and esters all contain a carbonyl group. (g) A compound with the molecular formula of C3H6O may be either an aldehyde, a ketone, or a carboxylic acid. (h) Bond angles about the carbonyl carbon of an aldehyde, a ketone, a carboxylic acid, and an ester are all approximately 109.5°. (i) The molecular formula of the smallest aldehyde is C3H6O, and that of the smallest ketone is also C3H6O. (j) The molecular formula of the smallest carboxylic acid is C2H4O2.arrow_forwardButanone is a four carbon compound with the functional group:(a) carboxylic acid(b) aldehyde(c) ketone(d) alcoholarrow_forwardGive the structural formulae and name the functional groups of the following compounds. (a) 3-chlorobut-1-ene Name the functional group: (b) butanedioic acid Name the functional group: (c) propanamide Name the functional group: (d) 3-methylbutanal Name the functional group:arrow_forward
- Draw the structure and name the product formed if the following alcohols are oxidized. Assume an excess of the oxidizing agent is used. If the alcohol is not expected to react with a chemical oxidizing agent, write NR (no reaction).(a) CH3CH2CH2CH2OH(b) 2-butanol(c) 2-methyl-2-propanol(d) 2-methyl-1-propanolarrow_forward(b) Differentiate between bioethanol and biodiesel.arrow_forward5.Write the structural formula of the ester that, when hydrolyzed, would yield the following:(a) methanol and propanoic acid(b) 1-octanol and acetic acid (c) ethanol and butanoic acidarrow_forward
- Draw a structural formula for each alcohol. (a) 2-Methyl-2-propyl-1,3-propanediol (b) 1-Octanol (c) 3,3-Dimethylcyclohexanolarrow_forwardWrite a condensed structural formula for each of the following:(a) an acid with the formula C4H8O2, (b) a cyclicketone with the formula C5H8O, (c) a dihydroxy compoundwith the formula C3H8O2, (d) a cyclic ester with theformula C5H8O2.arrow_forwardDraw the structures of the following carboxylic acids. (b) 2-bromobutanoic acidarrow_forward
- Write a condensed structural formula for a cyclic ester with the formula C5H8O2.arrow_forwardDraw the hydrogen bonding that takes place between(a) two molecules of ethanol.(b) two molecules of propylamine.(c) a molecule of dimethyl ether and two molecules of water.(d) two molecules of trimethylamine and a molecule of water.arrow_forwardWrite a balanced chemical equation using condensedstructural formulas for (a) the formation of butyl propionatefrom the appropriate acid and alcohol, (b) the saponification(base hydrolysis) of methyl benzoate.arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
