
Microbiology: An Introduction (13th Edition)
13th Edition
ISBN: 9780134605180
Author: Gerard J. Tortora, Berdell R. Funke, Christine L. Case, Derek Weber, Warner Bair
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5, Problem 2A
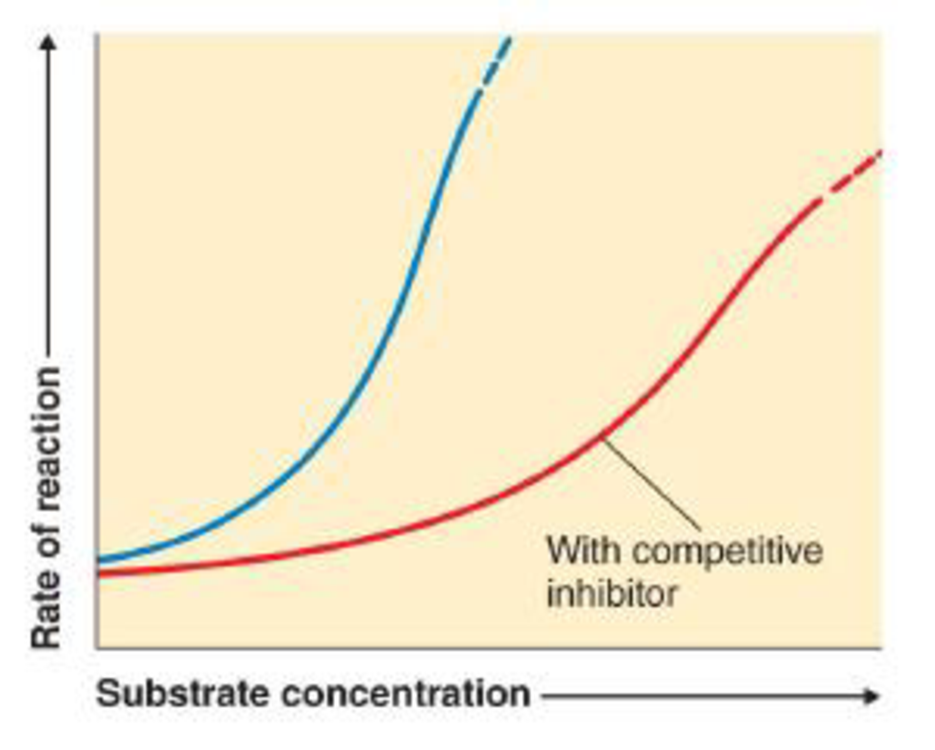
The following graph shows the normal
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
In the scheme below which represents the mechanism of action for a large number of enzymes:
A+B⟺AB⟶C
The steady state approximation is reached when:
d[AB]/dt≈0
k2≫k1
k−1≫k1
k−1=k1
An enzyme that catalyzes the reaction X ⇌ Y is isolated from two bacterial species. The enzymes have the same Vmax but different Km values for the substrate X. Enzyme A has a Km of 2.0 μM, and enzyme B has a Km of 0.5 μM. The plot below shows the kinetics of reactions carried out with the same concentration of each enzyme and with [X] = 1 μM. Which curve corresponds to which enzyme? Explain.
When studying the mechanism of the enzymatic reaction, functional groups were found that ensure the connection of the enzyme molecule with the substrate and take a direct part in the act of catalysis. What are these areas of the enzyme formed by these groups called? What functional structures form them and why?
Chapter 5 Solutions
Microbiology: An Introduction (13th Edition)
Ch. 5 - Prob. 1RCh. 5 - DRAW ITUsing the diagrams below, show each of the...Ch. 5 - DRAW IT An enzyme and substrate are combined. The...Ch. 5 - Define oxidation-reduction, and differentiate the...Ch. 5 - There are three mechanisms for the phosphorylation...Ch. 5 - All of the energy-producing biochemical reactions...Ch. 5 - Fill in the following table with the carbon source...Ch. 5 - Write your own definition of the chemiosmotic...Ch. 5 - Why must NADH be reoxidized? How does this happen...Ch. 5 - NAME IT What nutritional type is a colorless...
Ch. 5 - Which substance in the following reaction is being...Ch. 5 - Which of the following reactions produces the most...Ch. 5 - Prob. 3MCQCh. 5 - Which of the following compounds has the greatest...Ch. 5 - Prob. 5MCQCh. 5 - Prob. 6MCQCh. 5 - Which culture produces the most lactic acid? Use...Ch. 5 - Which culture produces the most ATP? Use the...Ch. 5 - Which culture uses NAD+? Use the following choices...Ch. 5 - Which culture uses the most glucose? Use the...Ch. 5 - Explain why, even under ideal conditions,...Ch. 5 - The following graph shows the normal rate of...Ch. 5 - Compare and contrast carbohydrate catabolism and...Ch. 5 - How much ATP could be obtained from the complete...Ch. 5 - The chemoautotroph Acidithiobacillus can obtain...Ch. 5 - Haemophilus influenzae requires hemin (X factor)...Ch. 5 - The drug Hivid, also called ddC, inhibits DNA...Ch. 5 - The bacterial enzyme streptokinase is used to...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Consider the experiment described in Section 2.1 in which Ted Garland and colleagues bred mice to run long dist...
Evolutionary Analysis (5th Edition)
What is the difference between histology and radiography?
Human Anatomy (8th Edition)
Nursing Student with Neuropathic Pain
Tamara Costa broke her right tibia and has undergone two separate surger...
Human Anatomy & Physiology (11th Edition)
Describe the evolution of mammals, tracing their synapsid lineage from early amniote ancestors to true mammals....
Loose Leaf For Integrated Principles Of Zoology
Relative thickness of the myocardium in different chambers; the functional significance of those differences; a...
Anatomy & Physiology: The Unity of Form and Function
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- The fractional saturation of an enzyme is 80% at a substrate concentration of 20 mM. In the presence of 5 mM of a competitive inhibitor the fractional saturation at the same substrate concentration is 40%. The Km for this reaction is mM, and the apparent Km is mM. The value of alpha is while the Ki of the inhibitor is mM.arrow_forwardThe figure displays the relationship between initial rate of product formation and reactant concentration in an enzyme-catalyzed reaction with a fixed amount of enzyme. Which of the following statements best explains the shape of the rate curve at high reactant concentration?arrow_forwardAn enzymes catalyzed reaction is studied in the presence and absence of an inhibitor. The following data was obtained in the image provided. Plot 1/[S] as abscissa and 1/V as ordinate for both catalyzed reactions and reaction with inhibitor. Use the same graph for both plots Calculate the following: Km of enzyme in the reaction without inhibitor Km' of the enzyme in the reation with inhibitor Vmax of the uninhibited reaction Vmax of the inhibited reaction What kind of inhibitor was added to the enzyme catalyzed reaction? Explain your answer in terms of changes in Km and Vmax.arrow_forward
- In an enzyme-catalyzed reversible reaction what happens when a) rate of change of enzyme-substrate complex concentration with time is positive b) rate of change of enzyme-substrate complex concentration with time is zero 9:0arrow_forwardIn an enzymatic reaction of maltase, the disaccharide maltose is hydrolyzed to glucose. In an experiment, an inhibitor X was added to the reaction. The addition of the inhibitor brought about a change in the activity of the enzyme. When the substrate concentration was 0.125 mM, the initial velocity was 0.10 mM per minute. However, Vo / Initial velocity changed to 0.25 mM per minute when the substrate concentration was 0.50 mM. Hence, calculate the of the reaction.arrow_forwardMolecule A is the substrate for enzyme X. Which is more likely to be a competitive inhibitor of enzyme X: molecule B or molecule C? Explain.arrow_forward
- An experiment on enzyme-catalyzed reaction was conducted in the laboratory by a student. Results obtained are summarized in the table below. In all the experiments, the concentration of the enzyme is the same. Substrate Concentration Velocity (pmol/min) (pmol) 1.5 0.21 3 0.28 4 0.32 0.36 8 0.4 15 0.45 18 0.47 1. Plot or graph these results using the Lineweaver-Burk method. 2. Determine the KM and Vmax values. Show all equations and calculations.arrow_forwardA researcher has measured the initial rate of an enzyme-catalyzed reaction as a function of substrate concentration in the presence and absence of 0.001μM inhibitor. She obtains the following data. What is the Vmax and Km for the "no inhibitor" and "+ inhibitor" experiments. Ans, What would be value of V0 for the "+inhibitor" at [S] = 0.4μM and the inhibitor is most likely a competitive, uncompetitive or mixed inhibitor.arrow_forwardThe graphs 3 and 4 representing 1/Vo = f(1/[S]o) have been done in the presence of a competitive (CI) and noncompetitive inhibitor (NCI). a- For each figure, determine from the relative position of the straight lines which one is obtained in presence of an inhibitor. b- Indicate which graph corresponds to the competitive inhibition and which one the noncompetitive inhibition. Justify your answer. c- Complete the graphs by indicating which values can be determined from the arrows. 3 1/No 1/[S]⁰ (4) 1/No 1/[S]oarrow_forward
- An enzyme catalyzes the following reaction. Which of the following inhibitors would you expect to be competitive inhibitors and which non-competitive inhibitors? Please explain briefly.arrow_forwardThe following reaction coordinate diagram charts the energy of a substrate molecule (S) as it passes through a transition state (X‡) on its way to becoming a stable product (P) alone or in the presence of one of two different enzymes (E1 and E2). How does the addition of either enzyme affect the change in Gibbs free energy (ΔG) for the reaction? Which of the two enzymes binds with greater affinity to the substrate? Which enzyme better stabilizes the transition state? Which enzyme functions as a better catalyst?arrow_forwardConsider an enzyme-catalyzed reaction that follows Michaelis-Menten kinetics with KM =3.0 mmol·dm-3. What concentration of a competitive inhibitor with KI = 20 umol·dm-3 willreduce the rate of formation of product by 50% when the substrate concentration is held at 0.10mmol·dm-3.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
Enzyme Kinetics; Author: MIT OpenCourseWare;https://www.youtube.com/watch?v=FXWZr3mscUo;License: Standard Youtube License