
Concept explainers
(a)
Interpretation:
The second resonance structure of the following ion should be drawn:
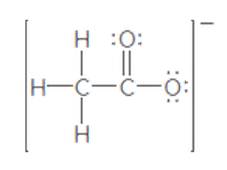
Concept Introduction:
When more than one Lewis structure can be drawn for a molecule or ion, then it is said to have resonance.
It is generally the delocalization of electrons (bonds) over three or more atoms in a molecule for which one simple Lewis structure cannot depicts its correct structure.
(b)
Interpretation:
The second resonance structure of the following ion should be drawn:
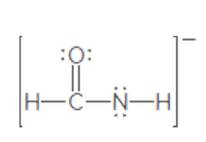
Concept Introduction:
When more than one Lewis structure can be drawn for a molecule or ion, then it is said to have resonance.
It is generally the delocalization of electrons (bonds) over three or more atoms in a molecule for which one simple Lewis structure cannot depicts its correct structure.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Draw a resonance structure that is NOT a proper resonance structure of the following ion?arrow_forward(a) Draw a second resonance structure for A. (b) Why can't a second resonance structure be drawn for B?arrow_forwardDrawing Resonance Structures Draw two more resonance structures for each species.arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
