
Concept explainers
(a)
Interpretation:
The indicated bond angles needs to be determined in the following compound:
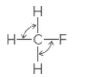
Concept Introduction:
The geometry around an atom depends on the number of bond pairs and lone pairs of electrons around it. There are approximate bond angles between atoms, defined for a particular geometry as follows:
| Molecular geometry around interested atom | Bond angle (') |
| Linear | 180 |
| Trigonal planer | 120 |
| Tetrahedral | 109.5 |
| Trigonal pyramidal | 109.5 |
| Bent | 109.5 |
(b)
Interpretation:
The indicated bond angles needs to be determined in the following compound:
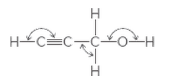
Concept Introduction:
The geometry around an atom depends on the number of bond pairs and lone pairs of electrons around it. There are approximate bond angles between atoms, defined for a particular geometry as follows:
| Molecular geometry around interested atom | Bond angle (') |
| Linear | 180 |
| Trigonal planer | 120 |
| Tetrahedral | 109.5 |
| Trigonal pyramidal | 109.5 |
| Bent | 109.5 |
(c)
Interpretation:
The indicated bond angles needs to be determined in the following compound:
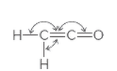
Concept Introduction:
The geometry around an atom depends on the number of bond pairs and lone pairs of electrons around it. There are approximate bond angles between atoms, defined for a particular geometry as follows:
| Molecular geometry around interested atom | Bond angle (') |
| Linear | 180 |
| Trigonal planer | 120 |
| Tetrahedral | 109.5 |
| Trigonal pyramidal | 109.5 |
| Bent | 109.5 |
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
General, Organic, and Biological Chemistry - 4th edition
- 2. Infer In a line-angle formula, each line represents a carbon-carbon bond. Each end of a line, as well as the intersection of lines, rep- resents a carbon atom. Knowing that carbon always forms four covalent bonds, explain how to determine the number of hydrogen atoms bonded to each carbon in a line-angle formula.arrow_forwardThere are at least three different molecules with the formula C3H8O. Draw a Lewis structure for each possible constitutional isomer. Be careful not to duplicate any structures.arrow_forward2. Identify the electron and molecular geometry of each indicated atoms in the compound below. HOarrow_forward
- For each the following molecules, indicate the direction of each bond polarity using arrows AND also indicate if the overall structure is polar or non- polar.arrow_forwardIndicate the number of single, double, and triple bonds in each of the three compounds. СH,O has single bonds, double bonds, and triple bonds. C,H, has single bonds, double bonds, and triple bonds. Со, has single bonds, double bonds, and triple bonds. careers prvacy po ig tems of use contact us belparrow_forwardDirections CHCЬ CH3CI CCla Write number of valence electrons for cach atom and find the total number of valence electrons Determine center atom and draw the skeleton Write the full Lewis Structure with all valence electrons Determine non-zero formal charges Center atom has regions Name the electronic geometry Center atom: # bonding atoms? # lone pairs? Name the molecular gcometry Draw the molecular geometry Identify polar covalent pairs. & Add dipole arrows for each polar bond Is there a net dipole? Is the molecule Polar or Nonpolar?arrow_forward
- Predict the indicated bond angles in each compound drawn as a Lewis structure with no implied geometry.arrow_forwardDraw a valid Lewis structure for each species. CH3CH3 CH5N C2H5Brarrow_forwardDirections NO. SO2 SO, Write number of valence electrons for cach atom and find the total number of valence electrons Determine center atom and draw the skeleton Write the full Lewis Structure with all valence electrons Determine non-zero formal charges Center atom has regions Name the electronic geometry Center atom: # bonding atoms? # lone pairs? Name the molecular geometry Draw the molecular geometry Identify polar covaient pairs. & Add dipole arrows for each polar bond Is there a net dipole? Is the molecule Polar or Nonpolar?arrow_forward
- Pick the approximate bond angle indicated of the molecule with the Lewis structure below. All atoms except H have an octet of electrons. Lone pairs are not indicated. H C-H H-C-0-C Harrow_forward2) NH3 a) How many lone pairs (non-bounding electron pairs) does the compound possess on All atoms? (central atom(s) and outer atoms? b) For this compound, Identify the following -number of electron groups (electron domains) -number of atoms bounded to the central atom -number of non-bounding electron pairs (lone pairs) attached to the central atom -General formula c) What is the Electron group Geometry also called "overall shape" and Hybrid Orbital designation for the central atm?arrow_forwardDraw VSEPR sketches for each compound below. Then, indicate whether the each of the following compounds is polar or nonpolar by drawing molecular dipole moments when appropriate. CHCl3 CH2Cl2 CCl4 H2O CH3CH2CH2CH2CH3arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning



