
Concept explainers
(a)
Interpretation:
The following pair should be labeled as resonance structures or not:
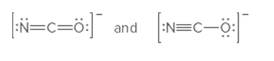
Concept Introduction:
When more than one Lewis structure can be drawn for a molecule or ion, then it is said to have resonance.
It is generally the delocalization of electrons (bonds) over three or more atoms in a molecule for which one simple Lewis structure cannot depicts its correct structure.
(b)
Interpretation:
The following pair should be labeled as resonance structures or not:
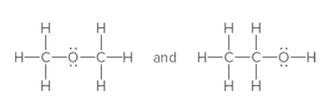
Concept Introduction:
When more than one Lewis structure can be drawn for a molecule or ion, then it is said to have resonance.
It is generally the delocalization of electrons (bonds) over three or more atoms in a molecule for which one simple Lewis structure cannot depicts its correct structure.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Rank the bonds according to increasing polarity. C-H N-H Si-H O-H Cl-Harrow_forwardWhich structure represents a correct resonance structure for the following structure? Select one:arrow_forwardConsider the resonance structures of formate. Select the true statements about the resonance structures. Each carbon-oxygen bond is somewhere between a single and double bond. Each oxygen atom has a double bond 50% of the time. The actual structure of formate is an average of the two resonance forms. The actual structure of formate switches back and forth between the two resonance forms. Incorrectarrow_forward
- Draw two resonance structures of the given molecule/ion. Be sure to include all lone pairs. :Ö:arrow_forwardConsider the resonance structures of formate. Select the true statements about the resonance structures. Each carbon-oxygen bond is somewhere between a single and double bond. The actual structure of formate is an average of the two resonance forms. Each oxygen atom has a double bond 50% of the time. The actual structure of formate switches back and forth between the two resonance forms.arrow_forwardPlace the bonds in order of increasing polar character. Place the bond with the least polar character at the top and the bond with the most polar character at the bottom.arrow_forward
- Draw 3 additional resonance structures that demonstrate which atoms have partial positive character.arrow_forwardDetermine what is wrong with each ionic Lewis structure and write the correct structure.arrow_forwardgive some tips how to find significant resonance structures of organic moleculesarrow_forward
- Consider the resonance structures of formate. ad Y Y Select the true statements about the resonance structures. The actual structure of formate is an average of the two resonance forms. Each carbon-oxygen bond is somewhere between a single and double bond. Each oxygen atom has a double bond 50% of the time. The actual structure of formate switches back and forth between the two resonance forms. Incorrectarrow_forwardDraw one valid Lewis structure for each compound. Assume the atoms are arranged as drawn.arrow_forwardExplain the differences among nonpolar covalent bonds, polar covalent bonds, and ionic bonds.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

