
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN: 9780618974122
Author: Andrei Straumanis
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 3, Problem 14CTQ
Interpretation Introduction
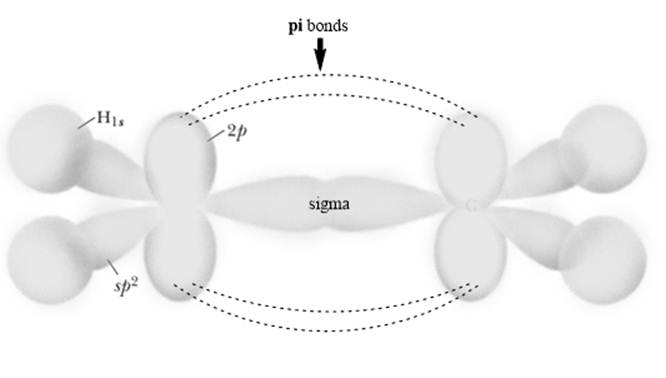
Interpretation: The reason behind K and L molecules as representatives of two different molecules but M and N representatives of same molecules should be given.
Concept introduction: Rotation about sigma bonds is feasible but however the rotation around pi bond is restricted because it disrupts the planarity of
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Need help with H, getting confused how many groups there is. I got 6 groups
Consider a molecule with the molecular formula C7H8O2 Draw a structure based on the data given.
( find structure asap plz C10H20 )
Chapter 3 Solutions
Organic Chemistry: A Guided Inquiry
Ch. 3 - Prob. 1CTQCh. 3 - What neutral atom is represented by the electron...Ch. 3 - Prob. 3CTQCh. 3 - Consider any one of the four identical hybrid...Ch. 3 - Prob. 5CTQCh. 3 - Prob. 6CTQCh. 3 - Prob. 7CTQCh. 3 - Prob. 8CTQCh. 3 - Prob. 9CTQCh. 3 - Prob. 10CTQ
Ch. 3 - On the left side of Figure 3.6, label the areas...Ch. 3 - Prob. 12CTQCh. 3 - Prob. 13CTQCh. 3 - Prob. 14CTQCh. 3 - Prob. 15CTQCh. 3 - Now consider the fully formed molecule on the...Ch. 3 - Prob. 1ECh. 3 - Explain why the two molecules below cannot...Ch. 3 - Prob. 3ECh. 3 - Consider the incomplete orbital representation of...Ch. 3 - Consider the following orbital representation of...Ch. 3 - Summarize how one determines the hybridization...Ch. 3 - Explain what is wrong with each of the following...Ch. 3 - Prob. 8ECh. 3 - Prob. 9ECh. 3 - Complete the following tables, and memorize their...Ch. 3 - Draw orbital representations of bonding in water...Ch. 3 - Draw electron configuration diagrams for carbon in...Ch. 3 - Prob. 13E
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- is there any line structure explanation of this?arrow_forwardChemistry 500 A Q2) 1 2 1 pick one. 2 3 4 All or none per row 1 to 7: (spelling counts) Monosodium glutamate (MSG) Center #electron regions (steric units) 5 6 3 S Legend: sodiumoxygen atom(red); carbon atom(black); nitrogen atom(blue); hydrogen atom(white). 7 Assignment #6 VSEPR large molecules: SEPR worksh... 6 #lone electron pairs ★, #bonded electron regions 4 electron geometry EG TE X # T 2023 500 Q6 V... molecular geometry MGH THE THUN Watch BUTTE var 100 M H 156 16 04105 wear ch Herale della COMEarrow_forwardDetermine a molecular formula, for example, CH4, from the line structure below. (Specify elements in the following order: C, H, others (in alphabetical order). Example: C4H7CIOS) Molecular formulaarrow_forward
- The problem is asking how many Sp3, Sp2, and Sp atoms there are and the answer key says 16 Sp3, 8 Sp2 and 2 Sp but I'm not even counting 26 carbons on this molecule so I'm a little confused.arrow_forwardFor each compound in the table below, decide whether there would be any hydrogen-bonding force between molecules of the compound, or between molecules of the compound and molecules of water. name dimethyl ether iodomethane acetic acid compound formula or Lewis structure H T H-C-Ö-C-H I H HILIA CH₂I | H H :O: | || H-C-C-O-H Between molecules of the compound? O yes O no O yes O no hydrogen-bonding force O yes O no Between molecules of the compound and molecules of water? O yes Ono O yes O no O yes O no X Sarrow_forwardThe so-called nitrogen rule states that if a compound has an odd number of nitrogen atoms, the value of m/z for its molecular ion will be an odd number. Why?arrow_forward
- Below each structure in the previous question is a “condensed structure” that tells you somethingabout how the atoms are arranged. Draw complete Lewis structures for each of the followingcondensed structures. (The net charge, if any, on each molecule is given at the end.) a. CH3CH2 b. CH2CH2 c. CH2CCH2 d. C( CH3)3+ e. BF4 f. NCO g. CH2OH+ (two different acceptable answers) h. CH2CHCHCHCH2+ which may alsobe written as CH2(CH)3CH2+ (more than one acceptable answer)arrow_forwardCheck the box next to each molecule on the right that has the shape of the model molecule on the left: molecules model (check all that apply) O COCI, | CH;0 You can c CH4 O CH,Cl, O None of the above Note for advanced students: the length of bonds and size of atoms in the model is not necessarily realistic. Th geometry and 3D shape of the molecule.arrow_forwardDraw a Lewis structure for the molecule below, showing all lone pairs. You may abbreviate any methyl groups as CH,. HOCH,CH,OCH; Click and drag to start drawing a structure.arrow_forward
- Consider the hydrocarbon HHH H H-C=Ċ-C-C=C-C-Harrow_forwardCalculate the IHD for each of the following molecular formulas. (a) C6H6; (b) C6H5NO2; (c) C8H13F2NO; (d) C4H12Si;(e) C6H5O-; (f) C4H6O3Sarrow_forwardDraw a Lewis structure for the molecule below, showing all lone pairs. You may abbreviate any methyl groups as CH,. CH,CH,OCH,CHCH, Click and drag to start drawing a structure.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning