
Interpretation:
The molecular formula, molecular weight and
Concept introduction:
The compounds having similar chemical formula but different structures are known as isomers.
The compounds having similar chemical or molecular formula but different connectivity is known as constitutional isomers.
Molecular formula of a compound contains
Molecular weight is defined as the addition of
Functional group is defined as the group of atoms or bonds present in a compound which is responsible for the
Answer to Problem 63A
The molecular mass and molecular weight of both glucose and fructose is same.
Fructose contains
Explanation of Solution
Glucose consists of single unit of sugar which can’t be hydrolyzed into simpler molecule. Glucose is obtained by the photosynthesis process and also found in plants.
Fructose consists of single unit of sugar which can’t be hydrolyzed into simpler molecule.
Glucose and fructose are structural isomers of each other implies they have same molecular formula that is
Molecular formula of glucose and fructose =
Molecular weight of glucose and fructose
=
=
Now, the structure of glucose and fructose is:
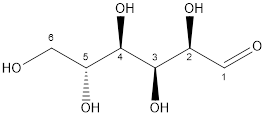
Glucose
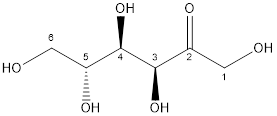
Fructose
Glucose and fructose both have six carbon atoms and five alcohol groups. But, fructose contains ketone (-C=O) functional group whereas glucose contains aldehyde (-CHO) functional group.
Chapter 23 Solutions
Chemistry: Matter and Change
Additional Science Textbook Solutions
Introductory Chemistry (6th Edition)
General, Organic, and Biological Chemistry (3rd Edition)
Inorganic Chemistry
Chemistry: The Central Science (13th Edition)
Chemistry: Structure and Properties (2nd Edition)
Chemistry: A Molecular Approach (4th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





