
Concept explainers
(a)
Interpretation:
The species with highest mass number among the given figure has to be given.
The given figure is,
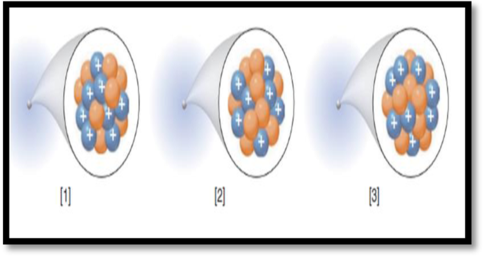
Figure 1
Concept Introduction:
Mass number (A):
Mass number of an element is defined as the total number of protons and neutrons in an atom.
(b)
Interpretation:
The number of electrons in each species given in the representation has to be given.
The given figure is,
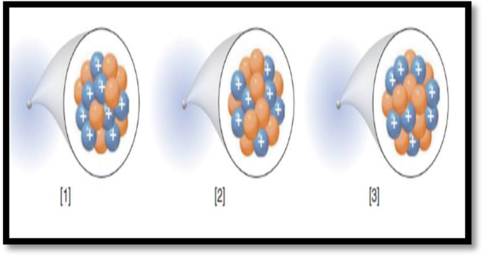
Figure 1
Concept Introduction:
Electrons:
Electrons have an electric charge of
(c)
Interpretation:
The isotopic symbol of each species in the given figure has to be given.
The given figure is,
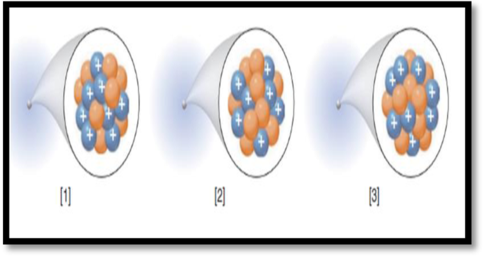
Figure 1
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Principles of General, Organic, Biological Chemistry
- Is it possible for the same Iwo elements to form more than one compound? Is this consistent with Dalton’s atomic theory? Give an example.arrow_forwardA cube of sodium has length 1.25 in. How many atoms are in that cube? (Note: dNa=0.968g/cm3.)arrow_forwardWhy is it important to understand atoms?arrow_forward
- Consider the chemical reaction as depicted below. Label as much as you can using the terms atom, molecule, element, compound, ionic, gas, and solid.arrow_forwardScientists J. J. Thomson and William Thomson (Lord Kelvin) made numerous contributions to our understanding of the atom’s structure. Which subatomic particle did J. J. Thomson discover, and what did this lead him to postulate about the nature of the atom? William Thomson postulated what became known as the “plum pudding” model of the atom’s structure. What did this model suggest?arrow_forwardClick on the site (http://openstaxcollege.org/l/16PhetAtomMass) and select the Mix Isotopes tab, hide the Percent Composition and Average Atomic Mass boxes, and then select the element boron. Write the symbols of the isotopes of boron that are shown as naturally occurring in significant amounts. Predict the relative amounts (percentages) of these boron isotopes found in nature. Explain the reasoning behind your choice. Add isotopes to the black box to make a mixture that matches your prediction in (b). You may drag isotopes from their bins or click on More and then move the sliders to the appropriate amounts. Reveal the Percent Composition and Average Atomic Mass boxes. How well does your mixture match with your prediction? If necessary, adjust the isotope amounts to match your prediction. Select Nature’s mix of isotopes and compare it to your prediction. How well does your prediction compare with the naturally occurring mixture? Explain. If necessary, adjust your amounts to make them match Nature’s amounts as closely as possible. 21. Repeat Exercise 2.20 using an element that has three naturally occurring isotopes.arrow_forward
- A titanium bicycle component has a volume of 32cm3. How many titanium atoms are in the bicycle component? Titanium density = 4.50g/cm3.arrow_forwardIn the following drawing, the green spheres represent atoms of a certain element. The purple spheres represent atoms of another element. If the spheres of different elements touch, they are part of a single unit of a compound. The following chemical change represented by these spheres may violate one of the ideas of Dalton’s atomic theory. Which one?arrow_forward

 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





