
Identify the elements used in each example of molecular art.

(a)
Interpretation:
The element in the molecular art has to be given.
The molecular art is,

Figure 1
Explanation of Solution
The red spheres in the above space filling figure 1 represents oxygen and the black spheres represent the carbon molecule. Thus, one particle contains two oxygen molecule and one particle contains one oxygen and one carbon molecule and another particle contains two oxygen and one carbon molecule.
(b)
Interpretation:
The element in the molecular art has to be given.
The molecular art is,
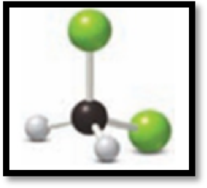
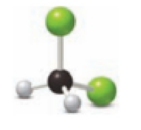
Figure 2
Explanation of Solution
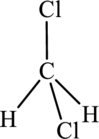
This ball and stick model represents dichloromethane. The molecule contains two Chlorine molecules (green balls), one carbon (black ball) and two hydrogen molecule (grey balls).
The structure of dichloromethane is,
Want to see more full solutions like this?
Chapter 2 Solutions
Principles of General, Organic, Biological Chemistry
- Give the systematic name for the following compounds that are found in everyday life: a. H2S (rotten egg smell) b. SO2 (smell of burnt matches) c. SFs (aerosol can propellant) d. Na2SO3 (dried fruit preservative)arrow_forwardLook around you and identify several objects that you think are probably made from polymers.arrow_forwardHow many metals are in the following groups? (a) Group 1 (b) Group 13 (c) Group 17arrow_forward
- When water boils, you can see bubbles rising to the surface of the water. Of what arc these bubbles made? air hydrogen and oxygen gas oxygen gas water vapor carbon dioxide gasarrow_forwardSketch a magnified view (showing atoms and/or molecules) of each of the following, and explain why the specified type of mixture is a heterogeneous mixture of two different compounds. a homogeneous mixture of an element and a compound.arrow_forwardRead The Molecular Revolution" box in this chapter on seeing atoms. How is the technology explained in the box a confirmation of the atomic theory? Does the technology "prove the atomic theory?arrow_forward
- a Which of the following substances would you expect to be elements and which would you expect to be compounds? 1 aluminum sulfate; 2 osmium; 3 radon; 4 lithium carbonate; 5 dimethylhydrazine. b On what general rule do you base your answers to part a? Can you name any exceptions to this general rule for compounds?arrow_forward2.94 Use a molecular level description to distinguish between LDPE and HDPE.arrow_forward
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





