
Concept explainers
(a)
Interpretation: The following reaction needs to be completed.
Concept Introduction : The reaction of
(a)
Answer to Problem 67E
Complete reaction is as follows:
Explanation of Solution
Fisher esterification reaction is as follows:
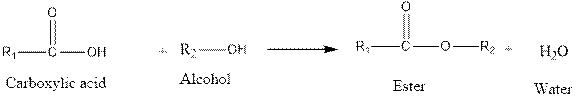
From the above reaction, it is clear that when carboxylic acid reacts with alcohol, an ester is formed along with water.
(b)
Interpretation: The following reaction needs to be completed.
Concept Introduction : The reaction of carboxylic acid with an alcohol in presence of acid catalyst produces an ester along with water. This type of reaction is termed as Fischer esterification reaction.
(b)
Answer to Problem 67E
Complete reaction is as follows:
Explanation of Solution
Fisher esterification reaction is as follows:
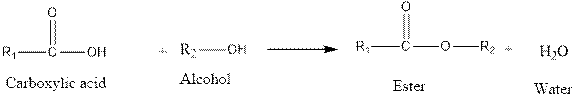
From the above reaction, it is clear that when carboxylic acid reacts with alcohol, an ester is formed along with water. In the given question carboxylic acid is formic acid. So, another carboxylic acid will form.
Want to see more full solutions like this?
Chapter 21 Solutions
Chemical Principles
- . The following chemical reactants produce the ester ethylethanoate (C4H8O2):C2H6O + CH3COOHWhat type of reaction occurs to make ethyl ethanoate?a. condensationb. hydrolysisc. combustiond. acid-base reactionarrow_forwardName the type of organic compound formed by each ofthe following reactions.a. elimination from an alcoholb. addition of hydrogen chloride to an alkenec. addition of water to an alkened. substitution of a hydroxyl group for a halogen atomarrow_forwardHO The IUPAC name for the line structural diagram shown above is O a. 1,5-pentanediol O b. pentane-1,5-diol O c. heptane-1,7-diol The organic compound shown in Question 7 is classified as a(n) O a. alcohol O b. carboxylic acid O c. ester Od. halogenated hydrocarbon The organic compound shown in Question 7 contains which functional group? O a. carboxyl O b. ester linkage O c. halogen Od. hydroxyl OH A student wrote the condensed formula shown below: CH₂CH₂COOH The IUPAC name for this condensed formula is: O a. propanol O b. ethanoic acid O c. propanoic acid O d. propanoate The organic compound in Question 10 is classified as a(n) Select one: O alcohol Oester O halogenated hydrocarbon O carboxylic acid The organic compound in Question 10 contains which functional group? Select one: O halogen O carboxyl Oester linkage O hydroxylarrow_forward
- OH 1. POCl3 pyridine 2. Cl₂ H₂O ? Major Organic Productarrow_forward1. what grouo does the ff organic compound belong? a. ketone b. ether c. cyloalkane d. esther 2. what group does the ff organic compound belong? a. amide b. azo c. nitrile d. amine 3. what is the priority functional group of the ff organic compound? a. carboxyl b. hydroxyl c. carbonyl d. hydroxidearrow_forward1. NaOCH,CH3 2. H3O* CH3CH2OHarrow_forward
- 4. Write the structure of the product resulting from the reaction of CH3 H,C- CH2arrow_forwardName the organic structure. H. CH2CH2CH2CH3 H.arrow_forwardThe addition of hydrogen (H2). using a catalyst such as Ni or Zn and heat and/or pressure will change an aldehyde into a primary alcohol an alkene into an alkane all of these an. alkyne into an alkane a ketone into a secondary alcoholarrow_forward
- Identify the organic functional groups and reaction type for the following reaction.The reactant is a(n)a. secondary alcohol and ketoneb. secondary alcohol and aldehydec. tertiary alcohol and ketoned. primary alcohol and ketonee. ether and carboxylic acidf. tertiary alcohol and aldehydeg. primary alcohol and aldehydeThe product is a(n)a. carboxylic acidsb. ketone and carbioxylic acidc. ether and carboxylic acidd. ketone and aldehydee. secondary alcohol and carboxylic acidf. secondary alcohol and ketoneThe reaction type is:a. hydrolysisb. reduction (hydrogenation)c. esterificationd. dehydratione. hydrationf. oxidationg. amide synthesisarrow_forwardThe functional group R carboxylic acid anhydrides. esters. aldehydes. ketones. amides. carboxylic acids. -0-C-R is characteristic ofarrow_forwardDraw the structure of each carboxylic acid or ester. a. pentanoic acid b. methyl hexanoate c. 3-ethylheptanoic acid d. butyl ethanoatearrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





