
(a)
Interpretation:
The suitable elements should be identified for given electronic configuration.
Concept introduction:
The periodic table is given in figure 1,
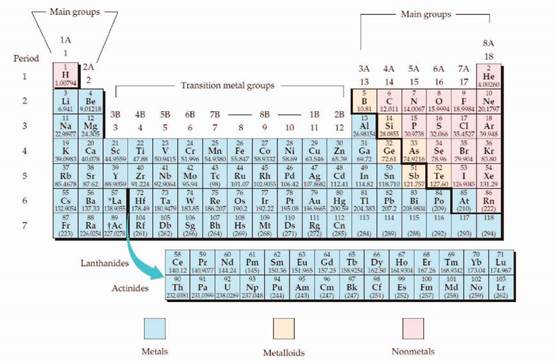
Figure 1
The electron configuration is the distribution of electrons of an atom or molecule in atomic or molecular orbitals. Atom consists of s, p, d, and f orbitals.
The Aufbau principle:
The Aufbau principle is states that in the ground state of an atom, electrons fill atomic orbitals of the lowest available energy levels before occupying higher levels. For example, the 1s shell is filled before the 2s subshell is occupied.
The subshell ordering by this rule is
(b)
Interpretation:
The suitable elements should be identified for given electronic configuration.
Concept introduction:
The periodic table is given in figure 1,
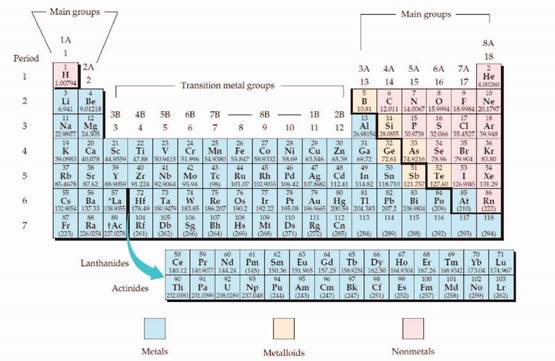
Figure 1
The electron configuration is the distribution of electrons of an atom or molecule in atomic or molecular orbitals. Atom consists of s, p, d, and f orbitals.
The Aufbau principle:
The Aufbau principle is states that in the ground state of an atom, electrons fill atomic orbitals of the lowest available energy levels before occupying higher levels. For example, the 1s shell is filled before the 2s subshell is occupied.
The subshell ordering by this rule is
(c)
Interpretation:
The suitable elements should be identified for given electronic configuration.
Concept introduction:
The periodic table is given in figure 1,
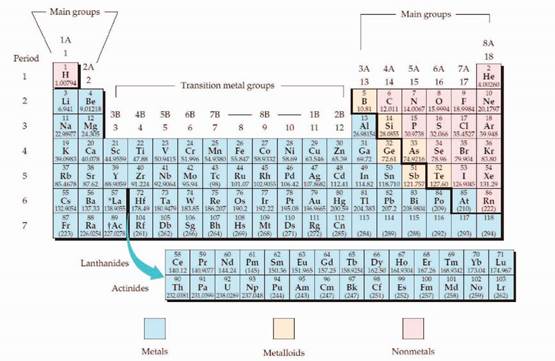
Figure 1
The electron configuration is the distribution of electrons of an atom or molecule in atomic or molecular orbitals. Atom consists of s, p, d, and f orbitals.
The Aufbau principle:
The Aufbau principle is states that in the ground state of an atom, electrons fill atomic orbitals of the lowest available energy levels before occupying higher levels. For example, the 1s shell is filled before the 2s subshell is occupied.
The subshell ordering by this rule is
(d)
Interpretation:
The suitable elements should be identified for given electronic configuration.
Concept introduction:
The periodic table is given in figure 1,
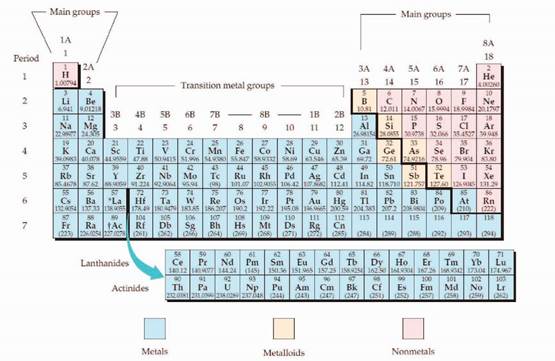
Figure 1
The electron configuration is the distribution of electrons of an atom or molecule in atomic or molecular orbitals. Atom consists of s, p, d, and f orbitals.
The Aufbau principle:
The Aufbau principle is states that in the ground state of an atom, electrons fill atomic orbitals of the lowest available energy levels before occupying higher levels. For example, the 1s shell is filled before the 2s subshell is occupied.
The subshell ordering by this rule is
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- Some different views of an octahedral symmetry metal complex 1 1 3 6. 2 1 4 View down a C, axis View down a C, axis View down a C,axis E 8C, 6C, 6C, 3C, i On i 6S, 85, 30, 604 A1g 1 1 1 1 1 1 1 1 1 -1 -1 1 Az8 1 1 -1 -1 1 Eg 2 z², x² - y? -1 0. -1 -1 -1 1 -1 -1 (xy, xz, yz) T2g -1 -1 1 1 -1 -1 A1u 1 1 1 -1 -1 -1 -1 A2u 1 -1 -1 1 -1 -1 -1 1 E -2 1 -2 -1 2 (x, y, z) -1 1 -1 -3 -1 1 3 T2u 1 -1 -1 -3 1 -1 The octahedral character table above may be useful for the following questions. 1. 1. 3. 3. 1. 2. 3. 1. 2)arrow_forwardCompound P was discovered by a scientist. Compound P is a dipeptide, optically active and has the molecular formula C„H14N2O3. Compound P is formed when compound Q and compound R joined together by condensation reaction. While, monomers S and T are formed by modifying the compounds Q and R. Polymer U is formed by the condensation reaction of monomers S and T. Draw the possible structural formulae of compounds P, Q, R, S, T and U. Label the peptide bond(s) for compound P. Draw the possible structural formulae for repeating unit of polymer U. Please state the number of functional groups present in compound P.arrow_forwardUsing the generic structure shown, indicate what the substituents are for the structure of finerenone (R1, R2, R3, for Ar indicate R6, R8, R9, R10, for D indicate R4). R1 R2 R3 = For Ar: R6 R8 = R9 = R10 = For D: R4= Finerenone: H₂N HC Generic formula: H₂N 230 R+ 'R'. R$ ofarrow_forward
- Figure 2.7 An atom may give, take, or share electrons with another atom to achieve a full valence shell, the most stable electron configuration. Looking at this figure, how many electrons do elements in group 1 need to lose in order to achieve a stable electron configuration? How many electrons do elements in groups 14 and 17 need to gain to achieve a stable configuration?arrow_forward1) Explain how g orbitals are different from s, p, d, and f atomic orbitals? - determine the number of g orbitals that will exist in a given energy level? - include a set of quantum numbers (n, l, and ml) for one of the g orbitals in the lowest energy set of g orbitals? 2) TlF3 forms a stable compound, while the similar compounds TlCl3 and TlBr3 are unstable and undergo disproportionation. Explain why the fluoro compound is more stable than the chloro and boromo compounds?arrow_forwardCompute the formal charge (FC) on each atom in the following structures [H2CNH2]+arrow_forward
- In which of the following pairs is the first element expected to have a higher electronegativity than the second? (1 Point) O O, P O Cs, Rb O I, Br Al, Parrow_forwardButadiene (right) is a colorless gas used to make synthetic rubber and many other compounds. (a) How many σ bonds and π bonds does the molecule have? (b) Are cis-trans arrangements about the double bonds possible? Explain.arrow_forwardI Review | Constants | Periodic Table You may want to reference (Page) Section 9.5 while completing this problem. Propane (C3 H8) burns in oxygen to produce carbon dioxide and water via the following reaction: C3HS (g) + 502 (g)→3CO2 (g) + 4H,0(g) Part A Calculate the mass of CO2 that can be produced if the reaction of 37.3 g of propane and sufficient oxygen has a 56.0 % yield. Express your answer with the appropriate units. l'i HẢ ? m(CO,) = Value Units Submit Request Answer Provide Feedback Next >arrow_forward
- The twisted proteins store elastic potential energy as it twists or untwists from its equilibrium state. The energy function for this system is shown in the figure where the energy is given in multiples of kBT which is a unit of energy used by biochemists when working at the molecular level. (1.0 kBT = 4.11×10−21 J). Figure 6 shows a horizontal line at an energy level of 136 kBT , which gives a total angular range of 2.095 radians. If the energy level of the ATP-synthase stalk is 100 kBT instead, between what angles is the stalk confined? Explain your reasoning.arrow_forwardFill in the blanks: Identify the oxidizing and reducing agent of the given equation: Answers should be the symbol of the element only. 8H*(aq) + 6Cl(aq) + Sn(s) + 4NO3-(aq) SnCl²(aq) + 4NO₂(g) + 4H₂O(1) 1. oxidizing agent 2. reducing agent = Sarrow_forwardDraw the Fischer projection formula for each of the following molecules. 1-36 а. b. ОН НО CH3 CI H i CH3 CH3 d. CH3 с. C. Cl ОН H CI НО Harrow_forward
 Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax

