
Concept explainers
Interpretation:
The reason should be explained for the third period in the periodic table contains eight elements.
Concept introduction:
The Aufbau principle:
The aufbau principle is states that in the ground state of an atom, electrons fill atomic orbitals of the lowest available energy levels before occupying higher levels. For example, the 1s shell is filled before the 2s subshell is occupied.
The subshell ordering by this rule is
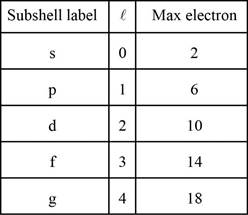
Number of electrons in the shells is given below,
Each shell contains only a fixed number of electrons. The first shell contains two electrons, the second shell contains eight (2 + 6) electrons, the third shell contains 18 (2 + 6 + 10), fourth shell contains (2 + 6 + 10 + 14 ) and so on.
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- Write the nuclear equation for the alpha decay of Po-214.arrow_forwardCompound A has molecular formula C7H7X. Its 1H-NMR spectrum shows a singlet at 2.25 ppm and two doublets, one at 7.28 ppm and one at 7.39 ppm. The singlet has an integral of three and the doublets each have an integral of two. The mass spectrum of A shows a peak at m/z = 126 and another peak at m/z = 128; the relative height of the two peaks is 3:1 respectively. Identify what atom X is, explaining your reasoning and identify Compound A, explaining your reasoning.arrow_forwardElement Z forms an ion Z 3+, which contains 31 protons. What is the identity of Z, and how many electrons does Z3+have?arrow_forward
- How many electrons must a nitrogen atom, Z = 7, gain to attain a noble gas configuration?arrow_forwardWhy do the 3s, 3p, and 3d orbitals have the same energy in a hydrogen atom but different energies in a many- electron atom?arrow_forwardWhy are the elements found in the I=P x A x T important in terms of them having an impact to our environment?arrow_forward
- For Be-10, find the: a.) mase defect (in grams) b.) binding energy in kilojoules per mole. mass proton= 1.00728 amu; mass neutron= 1.00867 amu; mass Be-10 = 10.013534679 amuarrow_forwardWrite the nuclear equation for the beta decay of Bi-214.arrow_forwardHow does the atomic structure or composition of the versions of sodium in question C above differ from a typical sodium atom, with its atomic mass of 23?arrow_forward
- Look up antimony in the periodic table 1Z = 512. How many covalent bonds would you expect it to form? Based on this information, which of the following antimony compounds is covalent and which is ionic: SbCl3 or SbCl5?arrow_forwardIf one of the planets in our solar system averages 0.723332 astronomical units from the sun, what is its orbital period (in Earth years, with one year = 365.256 Earth days)? the cubic root of (0.723332)1/2 = 0.947450 Earth years the square root of (0.723332)1/2 = 0.922220 Earth years the cubic root of (0.723332)1/3 = 0.964652 Earth years the square root of (0.723332)3 = 0.615186 Earth years the cubic root of (0.723332)2 = 0.805796 Earth yearsarrow_forward238. 234. 14) Uranium-238 ( 92") decays to form thorium-234 ( 90 h) with a half-life of 4.5 x 10° years. How many years will it take for 75% of the uranium-238 to decay? а) 9.0х 10 years с) 9.0х10° years b) 4.5 x 10° years d). 3.8 х 10° усаrsarrow_forward
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning

