
Interpretation:
The electronic configuration by shell and the metallic character of tin should be identified.
Concept introduction:
The periodic table is given in figure 1,
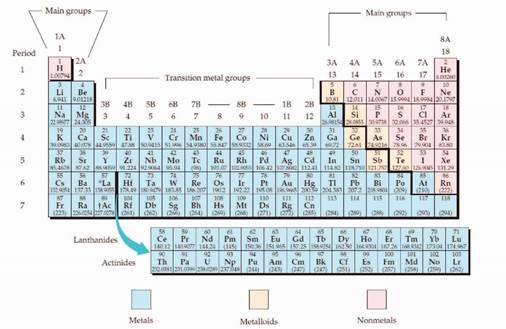
Figure 1
The Aufbau principle:
The aufbau principle is states that in the ground state of an atom, electrons fill atomic orbitals of the lowest available energy levels before occupying higher levels. For example, the 1s shell is filled before the 2s subshell is occupied.
The subshell ordering by this rule is
Number of electrons in the shells is given below,
Each shell contains only a fixed number of electrons. The first shell contains two electrons, the second shell contains eight
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- How many electrons are in the outer shell of each of the following atoms?arrow_forwardLook up the valence electron configuration, covalent atomic radius, effective nuclear charge, first ionization energy and Pauling electronegativity in Chapter 8 (tables are attached). Examine the above data and answer the following questions. a) Explain why some of the elements like TI and Pb on the lower left of the p block are metallic. b) Explain why some of the elements like C, Si in the center of the p block form covalent bonds. Explain why these bonds formed by the network of these elements (as studied in Chapter 25) tend to be unreactive. c) Explain why the noble Group 8A elements are highly unreactive gases. d) Explain why some elements like F, CI, Br etc, on the upper right of the p block are highly reactive nonmetals.arrow_forwardWhat if there are a different number of Neutrons? Sometimes elements have a different number of neutrons than usual, so therefore have a different atomic mass. This is what we call an Isotope (a variant form of an element which has the same number of protons but a differing number of neutrons). Sometimes we’ll see Isotopes labeled with their new mass like this: 14C = this is Carbon 14. Notice it has a different mass than what is on your periodic table The average mass of all the isotopes is defined as an element’s Atomic Weight. Can you answer questions about these Isotopes? ELEMENT ATOMIC MASS NUMBER OF PROTONS NUMBER OF NEUTRONS 14C A B C 15N D E F 2H G H I Use your table to look up the number of protons, remember that never changes!arrow_forward
- The atomic emission spectrum for a particular element includes blue-violet light with wavelength 440. nm. Calculate the energy in joules of this light given that E = h c/A, and h = 6.63 x 10-34Js, and c = 3.00 x 10°m/s. (h andc are constants, A is wavelength, convert nm into m) h c E =arrow_forwardCompound A has molecular formula C7H7X. Its 1H-NMR spectrum shows a singlet at 2.25 ppm and two doublets, one at 7.28 ppm and one at 7.39 ppm. The singlet has an integral of three and the doublets each have an integral of two. The mass spectrum of A shows a peak at m/z = 126 and another peak at m/z = 128; the relative height of the two peaks is 3:1 respectively. Identify what atom X is, explaining your reasoning and identify Compound A, explaining your reasoning.arrow_forwardWhat is the mass in grams of 6.022 * 1023 O atoms of mass 16.00 amu?arrow_forward
- Determine the mass in grams of 2.10 × 10²¹ atoms of copper. (The mass of one mole of copper is 63.55 g.)arrow_forwardWhat is the empirical formula for a compound that is 26.56% potassium, 35.41% chromium, and 38.03% oxygen?arrow_forwardArrange in order of increasing nonmetallic character (a) the period 3 elements Na, Cl, & Mg (b) the Group 7A elements At, F, & Iarrow_forward
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning

