
Interpretation:
Valance sub shell occupied by the electron should be identified for beryllium and arsenic atom.
Concept introduction:
The periodic table is given below,
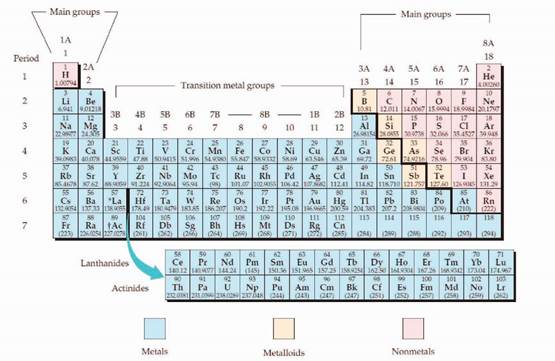
The electron configuration is the distribution of electrons of an atom or molecule in atomic or molecular orbitals. Atom consists of s, p, d, and f orbitals.
The Aufbau principle:
The Aufbau principle is states that in the ground state of an atom, electrons fill atomic orbitals of the lowest available energy levels before occupying higher levels. For example, the 1s shell is filled before the 2s subshell is occupied.
The subshell ordering by this rule is
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- Identify all of the chirality centers in the structure. The chirality centers are: A В C D `NH E F OH b H Он I J K L M N P Q Rarrow_forwardCircle the chiral carbons in the following compounds:arrow_forwardDetermine the mass in grams of 2.10 × 10²¹ atoms of copper. (The mass of one mole of copper is 63.55 g.)arrow_forward
- IDENTIFY THE FUNCTIONAL GROUP PRESENT IN THESE COMPOUNDS. A = ? B = ? C = ?arrow_forwardDetermine the mass in grams of 6.42 × 10²¹ atoms of carbon. (The mass of one mole of carbon is 12.01 g.)arrow_forwardIn which of the following pairs is the first element expected to have a higher electronegativity than the second? (1 Point) O O, P O Cs, Rb O I, Br Al, Parrow_forward
- . Give the name of and symbol for an element with this number of valence electrons.a) 2b) 6c) 8arrow_forwardIdentify the chirality center (sometimes called chiral atom) in each molecule. If the molecule does not contain a chirality center, select nonearrow_forwardThreonine has two chiral centers. Draw L-threonine and indicate which carbon atoms are chiral. Which carbon atom is responsible for d and L configuration?arrow_forward
- Draw the dipeptide that forms between alanine and glycine. Identify the net charge of this dipeptide at a pH = 0.5, 7.4, and 12.0.arrow_forwardIdentify the names of the following structure. (with alpha/beta and L-D designation)arrow_forwardHistidine has three ionizable groups. On the titration curve below, show: a) 2 molar equivalents of OH b) pka of the third ionizable group Histidine Titration 12.0 10.0 8.0 4 pH 6.0 4.0 2.0 0.0 Equivalents OHarrow_forward
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
