
(a)
Interpretation: The following compound should be classified according to its
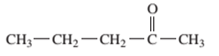
Concept Introduction: A group of atoms or an atom which is responsible for the characteristic reactions of a particular compound is said to be the functional group. Every functional group shows distinctive chemical properties irrespective to the moiety to which it is attached.
(b)
Interpretation: The following compound should be classified according to its functional group:
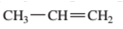
Concept Introduction: A group of atoms or an atom which is responsible for the characteristic reactions of a particular compound is said to be the functional group. Every functional group shows distinctive chemical properties irrespective to the moiety to which it is attached.
(c)
Interpretation: The following compound should be classified according to its functional group:
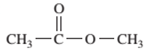
Concept Introduction: A group of atoms or an atom which is responsible for the characteristic reactions of a particular compound is said to be the functional group. Every functional group shows distinctive chemical properties irrespective to the moiety to which it is attached.
(d)
Interpretation: The following compound should be classified according to its functional group:
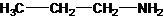
Concept Introduction: A group of atoms or an atom which is responsible for the characteristic reactions of a particular compound is said to be the functional group. Every functional group shows distinctive chemical properties irrespective to the moiety to which it is attached.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Basic Chemistry
- 12.52 Draw the condensed structural or line-angle formula for the alkene, aldehyde, or ketone product of each of the following reactions: (12.4) H, heat a. ОН CH; OH b. CHз — СH — СH-CH, OH [0] ОН d. CH3-CH2-CH2-CH-CH3arrow_forwardidulor 16.60 Draw the condensed structural formulas for the products from each of the following reactions: (16.2, 16.3) a. CH3-C-OH + NAOH → b. CH3-C-OH + H,O 7 ОН + КОН —arrow_forward12.58 Draw the condensed structural or line-angle formula, if cyclic, for each of the following: (12.3) a. formaldehyde c. 3-methyl-2-hexanone d. 3,5-dimethylhexanal b. 2-chlorobutanalarrow_forward
- (12.3)Which of the following has the strongest dispersion force between its molecules? O CH3CH3 O CH3CH₂CH₂CH₂CH3 O All of these have the dispersion forces with the same strength. O CH3CH₂CH₂CH3 O CH3CH₂CH₂CH₂CH₂CH3arrow_forward(12.9) Which of the following has the highest boiling point? O Both H₂S and H₂O have the same boiling point that is higher than the boiling point of H₂Se. O H₂Se O H₂O O H₂Sarrow_forward12.42 Which of the following will give a positive Tollens' test? (12.4) 1-propanol b. 2-propanol c. hexanal a.arrow_forward
- Give the iupac name of the following: 1. 2. 3. 4. CH3 CH=CH2arrow_forward4.15 Write the skeletal structure for the alkane or cycloalkane shown: a. C6H12 b. CH3CH₂CH₂CH3 c. CH3CH₂CH₂CH₂CH₂CH₂CH₂CH3arrow_forward11.10 Draw the condensed structural formula for alkanes or the line- angle formula for cycloalkanes for each of the following: ommo) emo2 to zslumot IS a. propane b. hexane c. heptane d. cyclohexane onstudolay the ends leboM onsqongolaarrow_forward
- 12.31 Write the IUPAC name for each of the following, using cis or trans prefixes, if needed: a. b. C. CH3-CH₂ H H H c=c C=C CH3 H H CH₂-CH₂-CH₂ - CH₂arrow_forward11.32 Draw the condensed structural formula for each of the following: b. trans-2-pentene cis-1,2-difluoroethene a. c. trans-2-heptenearrow_forwardKindly give the IUPAC names of the following alcohol compounds: 1. 2. 3. 4. 5. OH (CH₂)₂CHCH₂CH(CH₂CH₂CH(OH)CH₂CH₂ H₂CO LOCH, SHarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





