
General, Organic, and Biological Chemistry - 4th edition
4th Edition
ISBN: 9781259883989
Author: by Janice Smith
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17, Problem 67P
What products are formed when each ester is hydrolyzed with water and
a.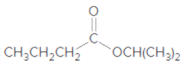
b.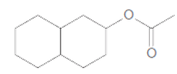
c.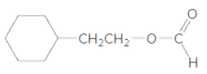
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
17. Which functional groups are in the following molecule of aspirin?
HO,
a benzene ring, an ester, a ketone, and an alcohol
b. a benzene ring, two ketones, an ether, and an alcohol
a benzene ring, a carboxylic acid, an ether, and a ketone
d. a benzene ring, a carboxylic acid, and an ester
a.
c.
Give the IUPAC name for each ketone.
C-CHCH,CH3
b.
a.
Does Phenols react with NaHCO3? Why or why not?
Chapter 17 Solutions
General, Organic, and Biological Chemistry - 4th edition
Ch. 17.1 - Draw out each compound to clearly show what groups...Ch. 17.1 - Prob. 17.2PCh. 17.1 - (a) Label each functional group in lisinopril, a...Ch. 17.2 - Give the IUPAC name for each compound. a. b. c.Ch. 17.2 - Give the structure corresponding to each IUPAC...Ch. 17.2 - Prob. 17.5PCh. 17.2 - Give an acceptable name for each ester. a. CH3(...Ch. 17.2 - Prob. 17.6PCh. 17.2 - Prob. 17.7PCh. 17.2 - Prob. 17.8P
Ch. 17.2 - Prob. 17.9PCh. 17.3 - Which compound in each pair has the higher boiling...Ch. 17.4 - Prob. 17.11PCh. 17.5 - In addition to ethyl butanoate (Section 17.5),...Ch. 17.6 - Prob. 17.3PPCh. 17.6 - Prob. 17.13PCh. 17.6 - Which compound ¡n each pair ¡s more water soluble?...Ch. 17.6 - Prob. 17.5PPCh. 17.6 - Prob. 17.14PCh. 17.6 - Prob. 17.15PCh. 17.7 - Ibuprofen is another pain reliever that is a...Ch. 17.8 - Prob. 17.6PPCh. 17.8 - Prob. 17.17PCh. 17.8 - Prob. 17.18PCh. 17.8 - Prob. 17.7PPCh. 17.8 - Prob. 17.19PCh. 17.9 - Prob. 17.8PPCh. 17.9 - Prob. 17.20PCh. 17.9 - Prob. 17.21PCh. 17.9 - Prob. 17.22PCh. 17.9 - Prob. 17.9PPCh. 17.9 - Prob. 17.23PCh. 17.9 - Prob. 17.24PCh. 17.10 - Prob. 17.10PPCh. 17.10 - Prob. 17.25PCh. 17.11 - Prob. 17.26PCh. 17 - Prob. 27PCh. 17 - Prob. 28PCh. 17 - Prob. 29PCh. 17 - Prob. 30PCh. 17 - Prob. 31PCh. 17 - Prob. 32PCh. 17 - Prob. 33PCh. 17 - Prob. 34PCh. 17 - Prob. 35PCh. 17 - Give an acceptable name for each ester. a. CH3CO2(...Ch. 17 - Prob. 37PCh. 17 - Prob. 38PCh. 17 - Prob. 39PCh. 17 - Prob. 40PCh. 17 - Prob. 41PCh. 17 - Prob. 42PCh. 17 - Prob. 43PCh. 17 - Draw the structure corresponding to each name. a....Ch. 17 - Draw the structure corresponding to each name. a....Ch. 17 - Draw the structure corresponding to each name. a....Ch. 17 - Prob. 47PCh. 17 - Prob. 48PCh. 17 - Prob. 49PCh. 17 - Prob. 50PCh. 17 - Prob. 51PCh. 17 - Prob. 52PCh. 17 - Prob. 53PCh. 17 - Prob. 54PCh. 17 - What ester is formed when butanoic acid...Ch. 17 - Prob. 56PCh. 17 - Prob. 57PCh. 17 - Prob. 58PCh. 17 - Prob. 59PCh. 17 - Prob. 60PCh. 17 - Prob. 61PCh. 17 - Prob. 62PCh. 17 - Prob. 63PCh. 17 - Prob. 64PCh. 17 - Prob. 65PCh. 17 - Prob. 66PCh. 17 - What products are formed when each ester is...Ch. 17 - Prob. 68PCh. 17 - Prob. 69PCh. 17 - Prob. 70PCh. 17 - Prob. 71PCh. 17 - Prob. 72PCh. 17 - Prob. 73PCh. 17 - Prob. 74PCh. 17 - Draw the products formed in each reaction. a. b....Ch. 17 - Draw the products formed in each reaction. a. b....Ch. 17 - Answer the following questions about A, depicted...Ch. 17 - Answer the following questions about B, depicted...Ch. 17 - Prob. 79PCh. 17 - Prob. 80PCh. 17 - Prob. 81PCh. 17 - Prob. 82PCh. 17 - Prob. 83PCh. 17 - Prob. 84PCh. 17 - Prob. 85PCh. 17 - Prob. 86PCh. 17 - Prob. 87PCh. 17 - Prob. 88PCh. 17 - Prob. 89CPCh. 17 - Lactams can be hydrolyzed with base, just like...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which is propyl propanoate? A. CH₂CH₂CH₂OOCCH₂CH; B. CH₂CH₂CH₂COOCH₂CH₂ C. CH₂CH₂CH₂COCH₂CH₂ D. CHỊCH,CH,OCH,CHỊCH, A B C Darrow_forward9. Label the following reactions as: A. ionization of a carboxylic acid in water B. neutralization of a carboxylic acid with a base C. formation of an ester D. acid-catalyzed hydrolysis of an ester E. ionization of an amine in water F. neutralization of an amine with an acid G. formation of an amide H. hydrolysis of an amide CH3NH₂ + HCI CH3NH3 Cl CH3CH₂CNHCH 3 HOH CH₂COH CH3COH+ NaOH →→→→ CHICOH heat CHICOH CH3COH + CH3NH₂ CH₂COH CH3CH₂COCH₂CH₂CH + HOHH CH3CH₂COH + HOCH₂CH₂CH3 CHO + H₂O - CH3CO heat CHO CH₂CO *Na + HOH CH3NH₂ + H₂0 CH3NH3* + OH- + H3O+ CH3CH₂C + NH2CH3 R-H R-O-H CH3CNHCH3 + HOH R-N- I O CH₂COH CH3COH + HOCH3 CH3COCH3 + HOH сно сосни, O || R-C-R O 11 R-C-O-R Hydrocarbons Alcohols Amines Ketones Esters R-X R-O-R 0=0|0=0|0=0 R-C-H R-C-0-H R-C-N- I Alkyl halides Ethers Aldehydes Carboxylic Acids Amidesarrow_forwardB. Esters Give the IUPAC name for each ester. CH3(CH2)4CO2CH3 CH3CH2CH2CH2COOCH2CH2CH3 CO₂CH2CH3 Draw the structure of each ester. Page 2 of 4 propyl propanoate butyl acetate methyl benzoatearrow_forward
- Why is it safe for us to consume foods like vinegar that contain acetic acids?arrow_forwardList the following compounds in order of increasing water solubility: a.ethoxyethane b.propanoic acid c.pentane d.1 butanolarrow_forwardGive the name of a carboxylic acid or carboxylate salt used in each of the following ways: a.As a soap b.As a general food preservative used to pickle vegetables c.As a preservative used in soft drinks d.As a treatment of athletes foot e.As a mold inhibitor used in bread. f.As a food additive noted for its pH buffering abilityarrow_forward
- What happens to phenol when: a) it reacts with Tollen's reagent? b) it undergoes esterification? c) it undergoes hydrolysis of esters?arrow_forward2. What carboxylic acid and alcohol are needed to prepare each ester by Fischer esterification? a) b) c)arrow_forwardDraw the structure of a compound tting each description:a. an aldehyde with molecular formula C4H8Ob. a ketone with molecular formula C4H8Oc. a carboxylic acid with molecular formula C4H8O2d. an ester with molecular formula C4H8O2arrow_forward
- Can an ester be synthesized from a carboxylic acid and alcohol in a 3 M sodium hydroxide solution? a. Yes, esters are synthesized in acid b. Yes, esters are synthesized in base c. No, esters are synthesized in base d. No, esters are synthesized in acidarrow_forwardDraw the structure of a constitutional isomer of 2-heptanone (CH 3COCH 2CH 2CH 2CH 2CH 3) that: a. contains an aldehyde b. contains a ketone c. contains a hydroxyl group (OH)arrow_forwardGive the IUPAC and common name for the following compounds. a. HO, b. но OHarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY