
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
5th Edition
ISBN: 9780321967466
Author: Karen C. Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16.1, Problem 16.7QAP
Draw the condensed structural formula for each of the following
a. 2-chloroethanoic acid
b. 3-hydroxypropanoic acid
c.-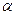 methylbutyric acid
methylbutyric acid
d. 3,5-dibromoheptanoic acid
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw structural formulas for the following carboxylic acids
and circle the acidic hydrogen atoms present:
a. pentanoic acid
b. 3,4-dichlorobutanoic acid
c. 3,3,5-trimethylhexanoic acid
Write an equation representing the neutralization of eachof the following carboxylic acids with KOH:a. 3-Chlorohexanoic acidb. Cyclohexanecarboxylic acidc. 3,4-Dimethylpentanoic acid
Draw the structure of each of the following carboxylic acids:a. b-Chlorobutyric acidb. a,b-Dibromovaleric acidc. b,g-Dihydroxybutyric acidd. d-Bromo-g-chloro-b-methylcaproic acid
Chapter 16 Solutions
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Ch. 16.1 - What carboxylic acid is responsible for the pain...Ch. 16.1 - What carboxylic acid is found in vinegar?Ch. 16.1 - Prob. 16.3QAPCh. 16.1 - Draw the condensed structural formula and give the...Ch. 16.1 - Prob. 16.5QAPCh. 16.1 - Prob. 16.6QAPCh. 16.1 - Draw the condensed structural formula for each of...Ch. 16.1 - Prob. 16.8QAPCh. 16.1 - Prob. 16.9QAPCh. 16.1 - Prob. 16.10QAP
Ch. 16.2 - Prob. 16.11QAPCh. 16.2 - Prob. 16.12QAPCh. 16.2 - Prob. 16.13QAPCh. 16.2 - Prob. 16.14QAPCh. 16.2 - Prob. 16.15QAPCh. 16.2 - Prob. 16.16QAPCh. 16.2 - Prob. 16.17QAPCh. 16.2 - Prob. 16.18QAPCh. 16.2 - Prob. 16.19QAPCh. 16.2 - Prob. 16.20QAPCh. 16.3 - Prob. 16.21QAPCh. 16.3 - Prob. 16.22QAPCh. 16.3 - Prob. 16.23QAPCh. 16.3 - Prob. 16.24QAPCh. 16.3 - Prob. 16.25QAPCh. 16.3 - Prob. 16.26QAPCh. 16.3 - 16.27 Give the IUPAC and common names,if any, of...Ch. 16.3 - Prob. 16.28QAPCh. 16.4 - Prob. 16.29QAPCh. 16.4 - Prob. 16.30QAPCh. 16.4 - Prob. 16.31QAPCh. 16.4 - Prob. 16.32QAPCh. 16.4 - Prob. 16.33QAPCh. 16.4 - Prob. 16.34QAPCh. 16.5 - Prob. 16.35QAPCh. 16.5 - Prob. 16.36QAPCh. 16.5 - Prob. 16.37QAPCh. 16.5 - Prob. 16.38QAPCh. 16.5 - Prob. 16.39QAPCh. 16.5 - Prob. 16.40QAPCh. 16 - Prob. 16.41UTCCh. 16 - Prob. 16.42UTCCh. 16 - Prob. 16.43UTCCh. 16 - Prob. 16.44UTCCh. 16 - Prob. 16.45UTCCh. 16 - Prob. 16.46UTCCh. 16 - Give the IUPAC and common names, if any, for each...Ch. 16 - Prob. 16.48AQAPCh. 16 - Prob. 16.49AQAPCh. 16 - Prob. 16.50AQAPCh. 16 - Prob. 16.51AQAPCh. 16 - Prob. 16.52AQAPCh. 16 - Prob. 16.53AQAPCh. 16 - Prob. 16.54AQAPCh. 16 - Prob. 16.55AQAPCh. 16 - Prob. 16.56AQAPCh. 16 - Prob. 16.57AQAPCh. 16 - Prob. 16.58AQAPCh. 16 - Prob. 16.59AQAPCh. 16 - Prob. 16.60AQAPCh. 16 - Prob. 16.61AQAPCh. 16 - Prob. 16.62AQAPCh. 16 - Prob. 16.63AQAPCh. 16 - Prob. 16.64AQAPCh. 16 - Prob. 16.65CQCh. 16 - Prob. 16.66CQCh. 16 - Prob. 16.67CQCh. 16 - Prob. 16.68CQCh. 16 - Prob. 16.69CQCh. 16 - Prob. 16.70CQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Write the condensed formula and the line formula foreach of the following carboxylic acids:a. 3-Methylhexanoic acidb. 2-Ethyl-2-methylpentanoic acidc. 3-Methylcyclopentanecarboxylic acidarrow_forwardThe IUPAC name for a carboxylic acid with three carbons in a straight chain would be A. propanoic acid B. ethylmethanoic acid C. propanalic acidarrow_forward14. The hydroxamic reaction involves medicinal substances that include: A. Ester group B. Primary aromatic group C. Amide group D. Aldehyde grouparrow_forward
- 10. Draw a structural formula for each of the fol 'owing esters: a. cyclohexyl propanoate b. methyl formate c. ethyl benzoate d. isopropyl acetate e. butyl butanoate f. propyl pentanoatearrow_forward16-18 Draw a condensed structural formulá that corresponds to each of the following carboxylic acids. a. 3,3-Dimethylheptanoic acid h. 4-Methylpentanoic acid c. 3-Chloropropanoic acid d. Trichloroethanoic acid 16.10arrow_forwardMestranol, sold under the brand names Enovid, Norinyl, and Ortho-Novum among others, is an estrogen medication which has been used in birth control pills. Select all of the functional group families, to which mestranol belongs. Choose one or more: A. Ester B. Alkene C. Ether D. Thiol E. Alcohol F. Arene G.Aldehyde H.Ketone I. Alkynearrow_forward
- The carboxylic acid compound is widely used as an antifungal agent a. oxalic b. benzoic c. salicylic d. aceticarrow_forwardDraw structures corresponding to the following: A. 2,3-dimethylhexanoic acid B. 4-methylpentanoic acid C. o-Hydroxybenzoic acid D. trans-Cyclobutane-1, 2-dicarboxylic acidarrow_forwardDraw each of the following using condensed formulas andline formulas:a. Ethyl isopropyl ketone c. Dibutyl ketoneb. Ethyl propyl ketone d. Heptyl hexyl ketonearrow_forward
- Select all of the intermolecular forces that are present in propanoic acid. (Carboxylic acids end with -COOH) 1. Hydrogen Bonding 2. Dipole-Dipole Forces 3. London Dispersion Forcesarrow_forwardDraw all possible the structural formula and give the IUPAC name of the following compounds:a. trinitrophenol b. dichlorobenzoic acid c. bromoanthracenearrow_forwardWORKSHEET No. 10 CARBOXYLIC ACID AND ESTERS NOMENCLATURE OF ESTER Complete the table below: PART Structure IUPAC Name A 1 1,3-Dimethyl Oxacyclobutane 2 3. 3-pentoxyoctane 4. 4-propoxyheptane 5. 6 7 8 1,3-dimethoxypropanearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY