
Interpretation:
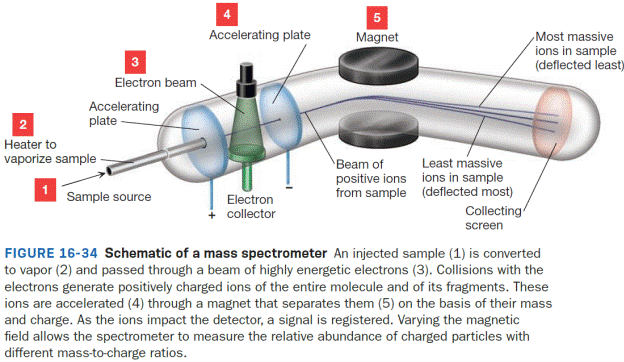
In Figure 16-34, a line to represent the path of an ion that is too light to reach the detector is to be drawn and labeled as “too light.” The same is to be done for an ion that is too massive to reach the detector and to be labeled as “too massive.”
Concept introduction:
Mass spectrometry provides insight into the mass of a molecule and the fragments that compose it. Unlike spectroscopy, however, mass spectrometry does not involve
Once vaporized, these gaseous molecules, M (g), drift through a beam of fast moving electrons. When an electron from that beam impacts a molecule of M (g), an electron from M (g) is knocked off, producing a gaseous species that has one fewer electron and therefore has a positive charge. The process of producing this molecular ion, M+ (g), is called electron impact ionization and can be represented by Equation 16-6.
A charged species like M+ (g) can be guided into a detector through a curved tube using a magnetic field to bend the ion’s path. The extent that a given ion’s path is bent depends on the strength of the magnetic field that is applied and the mass-to-charge ratio of the particle, represented as m/z. If the magnetic field strength is held fixed, then only ions of a specific value of m/z can reach the detector. Ions with m/z different from that value will collide with the wall of the tube and will be destroyed before reaching the detector; those that are too light will be deflected too much whereas those that are too massive will not be deflected enough. If multiple ions with different m/z values are present, then the magnetic field strength can be adjusted to allow the various ions to be detected. The detector itself is designed to keep track of the number of charged species that collide with it; this number is translated into what is called the relative abundance of the ion. The greater the number of charged species detected, the greater the ion’s relative abundance. If multiple ions are produced with different values of m/z, then the magnetic field strength can be scanned to determine the relative abundance at each m/z.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Mars is roughly 60 million km from the earth. How long does it take for a radio signal originating from the earth to reach Mars?arrow_forwardUsing the data given below, calculate the values of the neutron and proton separation energies for 37 Cl. 1 u = 931.478 MeV/ c² AM (36S) = -30.664 MeV AM (36CI) = -29.522 MeV AM (37CI) = -31.761 MeV [Z = 16 for Sulphur and Z= 17 for Chlorine.] mp = 938.28 MeV/c² mn = 939.57 MeV/c² me = 0.511 MeV/c²arrow_forward1- A red organic compound found in tomatoes that starts with the letter L? 2- Why is the visible spectrum a quantitative and not a qualitative analysis? 3- Why estimate cobalt using visible rays? And what kind of it?arrow_forward
- What is the frequency of radiation with a wavelength of 442 nm? What region of the EM spectrum would this light be in? Enter your answer here Save Answer 1arrow_forwardIf electrons are ejected from a given metal when irradiated with a 10-W red laserpointer, what will happen when the same metal is irradiated with a 5-W green laserpointer? (a) Electrons will be ejected, (b) electrons will not be ejected, (c) moreinformation is needed to answer this question.arrow_forwardUsing the GRSA(Given, Required, Solution, and Answer) Method solve the following: 1. What frequency is radiation with a wavelength of 5.00 x 10-6 cm? In what region of the electromagnetic spectrum is this radiation?arrow_forward
- A 0.1860 g rock sample containing an unknown amount of Ca was dissolved in HCI and diluted to 100.0 mL. Aliquots of 5.00 mL from this solution were added to different volumes of a 2.08 µg/mL. Ca standard solution and diluted to 200.0 mL. The emission intensity of these solutions was measured using atomic emission spectroscopy. The results are shown in the table. Unknown volume (mL) Standard volume (mL) Final volume (mL) Emission Intensity 0.00 200.0 5.00 200.0 10.00 200.0 15.00 200.0 20.00 200.0 Find the weight percent of Ca in the rock sample and its uncertainty using the method of standard additions. weight percent: 5.00 5.00 5.00 5.00 5.00 8.27 x10-5 Incorrect % Ca .03 Incorrect 227.0 413.0 637.0 831.01 1021.0arrow_forwardA metal whose threshold frequency is 2.07×10¹5 s-¹ emits an electron with a velocity of 7.03×105 m/s when radiation of 2.41x1015 s-¹ strikes the metal. Part A Use these data to calculate the mass of the electron. Express your answer with the appropriate units. m₂ = Submit μA Value Request Answer Units ?arrow_forwardIf a student determined the frequency of a line to be 3.456x1014 s-1 and this is value gives a percent error of 1.11% when compared to the true value, what is the true value for the frequency?arrow_forward
- Select the electron sink in H3O+. Keyboard controls (i Add selection ☆ + 0 ¡H Harrow_forward13.Which of the following is the reason that a more concentrated dye solution absorbs more light than a less concentrated solution? A. There are fewer molecules of dye in solution that can absorb electrons, meaning more electrons reach the detector, increasing absorbance. B. There are more molecules of dye in solution that can absorb electrons. C. There are more molecules of dye in solution that can absorb isotopes. D. There are more molecules of dye in solution that can absorb photons, meaning fewer photons reach the detector, increasing absorbance. E. There are fewer molecules of dye in solution that can absorb photons, meaning more photons reach the detector, increasing absorbance.arrow_forwardA 10.0077-g sample was analyzed for sodium by flame photometer and yielded the following data: If a sample gave an emission intensity of 3.07, what is the concentration of sodium, in μg/g, in the sample?arrow_forward

 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning




