
Concept explainers
(a)
Interpretation:
The number of signals that would be generated in the
Concept introduction:
In a
Answer to Problem 16.20P
The number of signals with their splitting patterns in the given molecule is indicated as
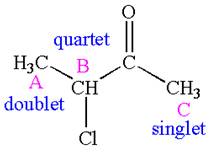
Explanation of Solution
The structure of the given compound is
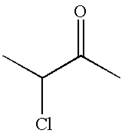
The compound has three chemically distinct protons indicated as
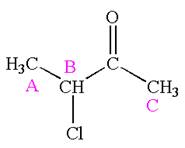
The protons
Splitting of each type of proton is shown below:
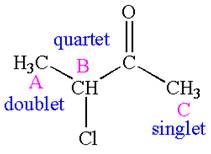
The splitting pattern for each type of proton signal in the given molecule is determined based on their respective number of neighboring protons.
(b)
Interpretation:
The number of signals that would be generated in the
Concept introduction:
In a
Answer to Problem 16.20P
The number of signals with their splitting patterns in the given molecule is indicated as
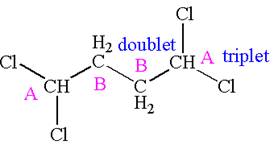
Explanation of Solution
The structure of the given compound is
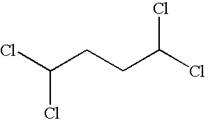
The compound has two chemically distinct protons indicated as
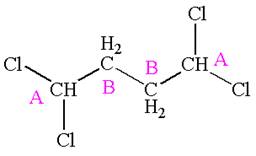
Protons
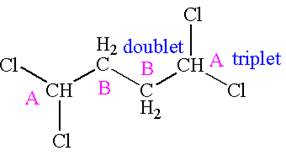
The splitting pattern for each type of proton signal in the given molecule is determined based on their respective number of neighboring protons.
(c)
Interpretation:
The number of signals that would be generated in the
Concept introduction:
In a
Answer to Problem 16.20P
The number of signals with their splitting patterns in the given molecule is indicated as
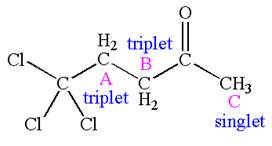
Explanation of Solution
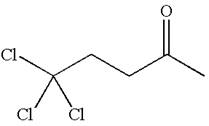
The structure of the given compound is
The compound has three chemically distinct protons indicated as
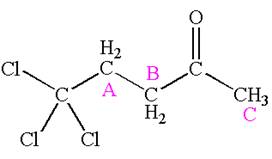
Protons

The splitting pattern for each type of proton signal in the given molecule is determined based on their respective number of neighboring protons.
(d)
Interpretation:
The number of signals that would be generated in the
Concept introduction:
In a
Answer to Problem 16.20P
The number of signals with their splitting patterns in the given molecule is indicated as
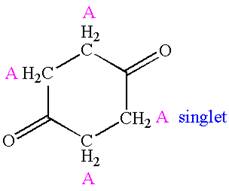
Explanation of Solution
The structure of the given compound is
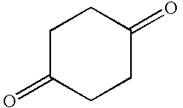
The compound has only one type of protons indicated, so there must be only one signal in
As all protons are chemically equivalent, they do not couple with each other’s signal and appear as singlet.

The splitting pattern for proton signal in the given molecule is determined based on their respective number of neighboring protons.
Want to see more full solutions like this?
Chapter 16 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Each of the following IR spectra is associated with one of the aromatic compounds below. Identify the compound associated with each spectrum. Any direction with explain or steps solve would be helpful!arrow_forwardDetermine the number of expected signals for the following compounds. (see picture attached).arrow_forwardWhich of the highlighted carbon atoms in each molecule absorbs farther downfield?arrow_forward
- How many peaks can you identify from the NMR spectrum? To which does each chemical shift peak corresponds?arrow_forwardMatch the compound to spectrum by drawing the structure next to correct spectrum. Then explain by describing the total number of signals each structure should have and which carbon would be farthest downfield.arrow_forwardMatch the spectra below to the molecule that most likely produces that spectra. Then, identify key absorption on the spectra.arrow_forward
- Which of the highlighted carbon atoms in each attached molecule absorbs farther downfield ?arrow_forwarda)Which of the compound in the picture best matches the following 1H NMR spectrum? Integration values are indicated next to their corresponding signal. Circle the answer. b) Briefly explain the answer (a) based on number of signals, position of signals and the integration of the signals.arrow_forwardReview Topica) (References) The chemical environment of protons can be deduced from their chemical shifts. What are the relative chemical shifts of the protons at the labeled positions? [Protons that are highly shielded have low delta (chemical shift) values.] 1) The proton with the lowest delta value (the most shielded) is The proton with the highest delta value (the most deshielded) is 2) The proton with the lowest delta value (the most shielded) is The proton with the highest delta value (the most deshielded) is Retry Entire Group 1 more group attempt remaining Submit Answerarrow_forward
- Based on the spectra you located, does your molecule have a carbonyl? If so, what functional group is it a part of (carboxylic acid, ketone, aldehyde, ester, amide) and what is the frequency (in wavenumbers) of the absorption peak? If not, what is the approximate frequency range for a carbonyl? Does your molecule have either an –O-H or –N-H bond? If so, what functional group is it a part of (carboxylic acid, alcohol, amine, amide) and what is the frequency (in wavenumbers) of the absorption peak? If not, what are the approximate frequency ranges for an –O-H and an –N-H bond? Does your molecule have either an alkyne or nitrile functional group? If so, which functional group is it and what is the frequency (in wavenumbers) of the absorption peak? If not, what is the approximate frequency range for a triple bond?arrow_forwardEach of the molecules shown below generates two different HNMR signals. Match each molecule with one of the HNMR spectra given below.arrow_forwardNext to each spectrum given below, determine which of the suggested compounds it belongs to.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

