
(a)
Interpretation:
Aldehyde must contain the element nitrogen or not has to be indicated.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom.
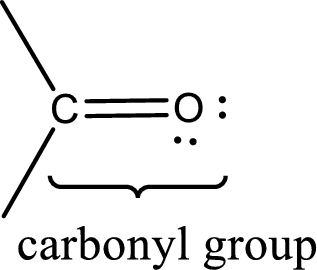
The groups that is attached to the carbonyl carbon atom can be either hydrogen or carbon atom. If the attached atoms are hydrogen and a carbon atom means then the compound is an aldehyde and if they are two carbon atoms means then the compound is a ketone. If the attached atom is a nitrogen atom to the carbonyl carbon atom means it is a amide. If a hydroxyl group is attached to a carbonyl carbon atom, then it is a
(b)
Interpretation:
Ketone must contain element nitrogen or not has to be indicated.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
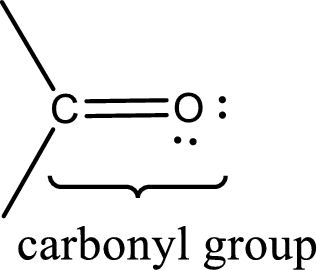
The groups that is attached to the carbonyl carbon atom can be either hydrogen or carbon atom. If the attached atoms are hydrogen and a carbon atom means then the compound is an aldehyde and if they are two carbon atoms means then the compound is a ketone. If the attached atom is a nitrogen atom to the carbonyl carbon atom means it is a amide. If a hydroxyl group is attached to a carbonyl carbon atom, then it is a carboxylic acid. If one of the bond in the carbonyl carbon atom is attached to an oxygen atom that is bonded with alkyl, cycloalkyl or aromatic system is known as ester.
(c)
Interpretation:
Ester must contain element nitrogen or not has to be indicated.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
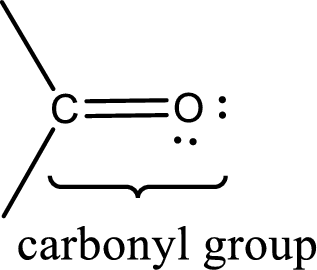
The groups that is attached to the carbonyl carbon atom can be either hydrogen or carbon atom. If the attached atoms are hydrogen and a carbon atom means then the compound is an aldehyde and if they are two carbon atoms means then the compound is a ketone. If the attached atom is a nitrogen atom to the carbonyl carbon atom means it is a amide. If a hydroxyl group is attached to a carbonyl carbon atom, then it is a carboxylic acid. If one of the bond in the carbonyl carbon atom is attached to an oxygen atom that is bonded with alkyl, cycloalkyl or aromatic system is known as ester.
(d)
Interpretation:
Amide must contain element nitrogen or not has to be indicated.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
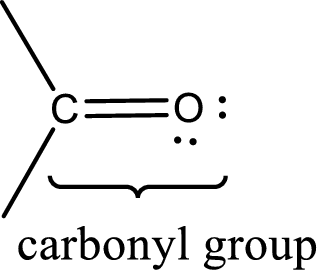
The groups that is attached to the carbonyl carbon atom can be either hydrogen or carbon atom. If the attached atoms are hydrogen and a carbon atom means then the compound is an aldehyde and if they are two carbon atoms means then the compound is a ketone. If the attached atom is a nitrogen atom to the carbonyl carbon atom means it is a amide. If a hydroxyl group is attached to a carbonyl carbon atom, then it is a carboxylic acid. If one of the bond in the carbonyl carbon atom is attached to an oxygen atom that is bonded with alkyl, cycloalkyl or aromatic system is known as ester.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
General, Organic, and Biological Chemistry
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





