
(a)
Interpretation:
Aldehyde must contain a carbonyl group or not has to be indicated.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom.
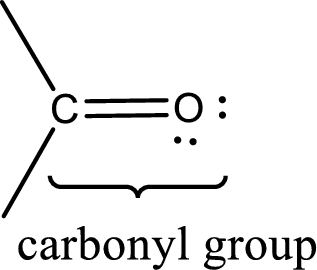
The groups that is attached to the carbonyl carbon atom can be either hydrogen or carbon atom. If the attached atoms are hydrogen and a carbon atom means then the compound is an aldehyde and if they are two carbon atoms means then the compound is a ketone. If the attached atom is a nitrogen atom to the carbonyl carbon atom means it is a amide. If a hydroxyl group is attached to a carbonyl carbon atom, then it is a
(b)
Interpretation:
Ketone must contain a carbonyl group or not has to be indicated.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
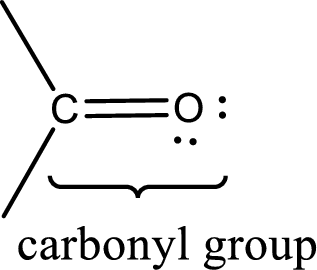
The groups that is attached to the carbonyl carbon atom can be either hydrogen or carbon atom. If the attached atoms are hydrogen and a carbon atom means then the compound is an aldehyde and if they are two carbon atoms means then the compound is a ketone. If the attached atom is a nitrogen atom to the carbonyl carbon atom means it is a amide. If a hydroxyl group is attached to a carbonyl carbon atom, then it is a carboxylic acid. If one of the bond in the carbonyl carbon atom is attached to an oxygen atom that is bonded with alkyl, cycloalkyl or aromatic system is known as ester.
(c)
Interpretation:
Amide must contain a carbonyl group or not has to be indicated.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
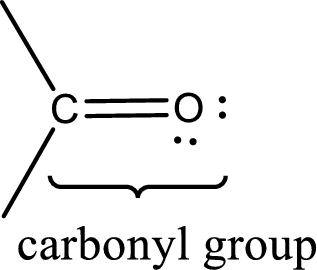
The groups that is attached to the carbonyl carbon atom can be either hydrogen or carbon atom. If the attached atoms are hydrogen and a carbon atom means then the compound is an aldehyde and if they are two carbon atoms means then the compound is a ketone. If the attached atom is a nitrogen atom to the carbonyl carbon atom means it is a amide. If a hydroxyl group is attached to a carbonyl carbon atom, then it is a carboxylic acid. If one of the bond in the carbonyl carbon atom is attached to an oxygen atom that is bonded with alkyl, cycloalkyl or aromatic system is known as ester.
(d)
Interpretation:
Carboxylic acid must contain a carbonyl group or not has to be indicated.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
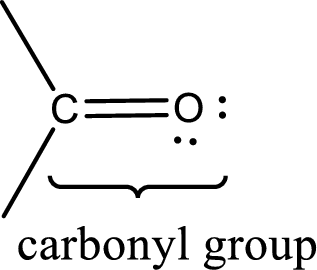
The groups that is attached to the carbonyl carbon atom can be either hydrogen or carbon atom. If the attached atoms are hydrogen and a carbon atom means then the compound is an aldehyde and if they are two carbon atoms means then the compound is a ketone. If the attached atom is a nitrogen atom to the carbonyl carbon atom means it is a amide. If a hydroxyl group is attached to a carbonyl carbon atom, then it is a carboxylic acid. If one of the bonds in the carbonyl carbon atom is attached to an oxygen atom that is bonded with alkyl, cycloalkyl or aromatic system is known as ester.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
General, Organic, and Biological Chemistry
- (12) Which one of the following statements about fluo- rometry is FALSE? a) Fluorescence is better detected at 90 from the exci- tation direction. b) Fluorescence is typically shifted to longer wave- length from the excitation wavelength. c) For most fluorescent compounds, radiation is pro- duced by a transitionarrow_forwardDon't used Ai solutionarrow_forwardDon't used Ai solutionarrow_forward
- Don't used Ai solutionarrow_forwardIndicate the correct option.a) Graphite conducts electricity, being an isotropic materialb) Graphite is not a conductor of electricityc) Both are falsearrow_forward(f) SO: Best Lewis Structure 3 e group geometry:_ shape/molecular geometry:, (g) CF2CF2 Best Lewis Structure polarity: e group arrangement:_ shape/molecular geometry: (h) (NH4)2SO4 Best Lewis Structure polarity: e group arrangement: shape/molecular geometry: polarity: Sketch (with angles): Sketch (with angles): Sketch (with angles):arrow_forward
- 1. Problem Set 3b Chem 141 For each of the following compounds draw the BEST Lewis Structure then sketch the molecule (showing bond angles). Identify (i) electron group geometry (ii) shape around EACH central atom (iii) whether the molecule is polar or non-polar (iv) (a) SeF4 Best Lewis Structure e group arrangement:_ shape/molecular geometry: polarity: (b) AsOBr3 Best Lewis Structure e group arrangement:_ shape/molecular geometry: polarity: Sketch (with angles): Sketch (with angles):arrow_forward(c) SOCI Best Lewis Structure 2 e group arrangement: shape/molecular geometry:_ (d) PCls Best Lewis Structure polarity: e group geometry:_ shape/molecular geometry:_ (e) Ba(BrO2): Best Lewis Structure polarity: e group arrangement: shape/molecular geometry: polarity: Sketch (with angles): Sketch (with angles): Sketch (with angles):arrow_forwardDon't used Ai solutionarrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





