
Concept explainers
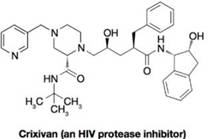
The problem below is directed toward devising a hypothetical pathway for the synthesis of the acyclic central portion of Crixivan (Merck and Company’s HIV protease inhibitor). Note that your synthesis might not adequately control the stereochemistry during each step, but for this particular exercise chat is not expected.
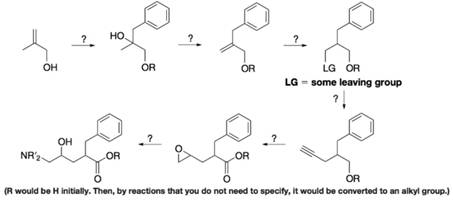
Fill in missing compounds and reagents in the following outline of a hypothetical synthesis of the acyclic central portion of Crixivan. Note that more than one intermediate compound may be involved between some of the structures shown below.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Chemistry For Changing Times (14th Edition)
Chemistry
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Chemistry & Chemical Reactivity
Chemistry
Introductory Chemistry (6th Edition)
- Show how Friedel–Crafts acylation might be used to synthesize the followingcompounds.(a) acetophenonarrow_forwardGive the structure of compounds A to F in the following series of reactionsarrow_forwardCompound A is first reacted with methylamine in the presence of acid and then treated with NaBH3CN. Using the spectroscopic data given, what is the structure of the product after step 1?arrow_forward
- (4) Provide the reagents necessary to carry out the following synthesis. + enantiomer (A) (1) (i) BH3, (ii) H₂O2, NaOH; (2) NaOEt JATOT (B) (1) (i) BH3, (ii) H₂O2, NaOH; (2) TsCl, pyridine; (3) KOC(CH3)3 (C) (1) H3O+, (2) TsCl, pyridine, (3) KOC(CH3)3 (D) (1) HBr (no peroxides); (2) NaOEtarrow_forwardBelow is a schematic representation of possible reactions that Compound X can undergo. Use the scheme to answer the following question. Illustrating with reaction mechanisms, show how compounds [1], [2], [3] and [4] are formed. The attached image contains the schemearrow_forwardThe Arndt-Eistert synthesis involves a rearrangement (known as a Wolff Rearrangement) with concomitant evolution of nitrogen gas. This reaction has been modified by Georg (J. Am. Chem. Soc. 2010, 132, 15512) to result in a "vinylogous" Wolff Rearrangement (not covered in class). Propose a mechanism for Georg's variation shown below (hint: there's a ketene intermediate). Bn. N O OEt №₂ Bn CO₂Etarrow_forward
- Propose a reasonable mechanism for the following reaction.arrow_forwardA ketone undergoes acid-catalyzed bromination, acid-catalyzed chlorination, racemization, and acid-catalyzed deuterium exchange at the alpha-carbon. All of these reactions have similar rate constants. What does this tell you about the mechanisms of these reactions?arrow_forwardprovide a possible synthesis for the following reactionarrow_forward
- Propose a reasonable reaction mechanism for the transformation given below and provide a brief explanationarrow_forwardTreatment of a cyclic ketone with diazomethane is a method for accomplishing a ring-expansion reaction. The reaction involves the initial nucleophilic attack by diazomethane on the carbonyl carbon to form a tetrahedral intermediate. Collapse of this intermediate is accompanied by bond migration and loss of N,. For example, treatment of cyclohexanone with diazomethane yields cycloheptanone. CH2N2, ether + N2 Draw the structure of the organic product(s) of the ring expansion of this compound: • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading. • Draw one structure per sketcher Add additional ketchers usina the dron down menu in the bottom right co Previous Nextarrow_forwardgas, Cl¬CH2CH2¬S¬CH2CH2¬Cl, was used as a poisonous chemical agentin World War I. Mustard gas is much more toxic than a typical primary alkyl chloride. Itstoxicity stems from its ability to alkylate amino groups on important metabolic enzymes,rendering the enzymes inactive.(a) Propose a mechanism to explain why mustard gas is an exceptionally potent alkylatingagent.(b) Bleach (sodium hypochlorite, NaOCl, a strong oxidizing agent) neutralizes and inactivates mustard gas. Bleach is also effective on organic stains because it oxidizes coloredcompounds to colorless compounds. Propose products that might be formed by thereaction of mustard gas with bleach.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

