
Concept explainers
Interpretation:
The mechanism of the given reaction is to be written.
Concept introduction:
The reaction of Grignard reagents with esters yields secondary and tertiary alcohols as their products. These reactions are two-step reactions. In the first step, nucleophilic addition of carbonyl group takes place because Grignard reagent being nucleophilic uses its lone pair of electrons to form a bond with a carbon atom. This results in the formation of an alkoxide ion that remains associated with
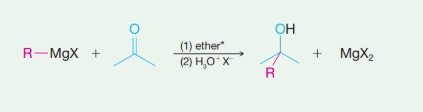
In the second step, addition of aqueous HX causes the protonation of the alkoxide ion which leads to the formation of the alcohol and
Carbonyl groups of cyclic esters react with 2 equivalents of Grignard reagents and followed by hydrolysis to produce tertiary alcohols.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Organic Chemistry
- Complete each acid-base reaction and predict whether the position of equilibrium lies toward the left or toward the right. (a) CH3CCH+CH3CH2ONa+CH3CH3OH (b) CH3CCCH2CH2OH+Na+NH2NH3(l)arrow_forwardDraw the structure(s) for the major final product(s) formed in the following reaction sequence. CN ༼་་ SO 503 H₂SO4 Cl FeCl3 Click and drag to start drawing a structure.arrow_forwardExplain why pentane-2,4-dione forms two alkylation products (A and B) when the number ofequivalents of base is increased from one to two.arrow_forward
- b) The Wolf-Kishner reduction is a reaction used in Organic Chemistry to convert carbonyl functionalities into methylene group. The reaction was used to convert an aldehyde or ketone to an alkane using hydrazine, base and thermal conditions. The mechanism begins with the attack of hydrazine of the aldehyde or ketone. Stage 1: The reaction of aldehyde/ketone with hydrazine to produce hydrazine Stage 2: Reaction with the base and heat to convert hydrozone to alkane Write the mechanism of the reaction.arrow_forwardProvide the mechanism for this reaction. Please show arrow movements, all line pairs, and formal chargesarrow_forward4) Give an example of an enol which would tautomerize into a) an aldehyde and b) a ketone.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


