
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12, Problem 21P
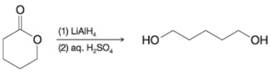
Write a mechanism for the following reaction. You may use H- to represent hydride ions from
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
It is a type of organic reaction in which parts from two molecules
exchange. *
Elimination
Substitution
Addition
Rearrangement
In the reaction given below, what type of addition reaction is used? *
H
H
H
H
H-C-c=C
HBr
н-с — с — С —с —н
+
H
H
H
Br
H
O Hydrogenation
O Hydrohalogenation
O Halogenation
O Dehalogenation
H-O-I
Example:
H
R
11' COO-
Ca
↓
NH3
L or D?
Choose a likely identity for X, Y, and Z in these structures.
O Be
Be
O B
ON
Be
O O O O O O
O O O O OC
O O O O OO
Chapter 12 Solutions
Organic Chemistry
Ch. 12 - Prob. 1PPCh. 12 - Prob. 2PPCh. 12 - Prob. 3PPCh. 12 - PRACTICE PROBLEM 12.4 Predict the products of the...Ch. 12 - Prob. 5PPCh. 12 - Prob. 6PPCh. 12 - Practice Problem 12.7
Provide retrosynthetic...Ch. 12 - Prob. 8PPCh. 12 - What products would you expect from the reaction...Ch. 12 - What products would you expect from the reaction...
Ch. 12 - What product (or products) would be formed from...Ch. 12 - Prob. 12PCh. 12 - 12.13 Write reaction conditions and the product...Ch. 12 - Prob. 14PCh. 12 - Predict the organic product from each of the...Ch. 12 - Predict the organic product from each of the...Ch. 12 - Predict the organic product from each of the...Ch. 12 - Predict the major organic product from each of the...Ch. 12 - Synthesize each of the following compounds from...Ch. 12 - Prob. 20PCh. 12 - 21. Write a mechanism for the following reaction....Ch. 12 - Prob. 22PCh. 12 - 23. What organic products A-H would you expect...Ch. 12 - Prob. 24PCh. 12 - Show how 1-pentanol could be transformed into each...Ch. 12 - Provide the reagents needed to accomplish...Ch. 12 - Prob. 27PCh. 12 - For each of the following alcohols, write a...Ch. 12 - Prob. 29PCh. 12 - Prob. 30PCh. 12 - Prob. 31PCh. 12 - Prob. 32PCh. 12 - Predict the major organic product from each of the...Ch. 12 - 34. Synthesize the following compound using...Ch. 12 - Prob. 35PCh. 12 - Prob. 36PCh. 12 - 37. Explain how and IR spectroscopy could be used...Ch. 12 - 38. An unknown X shows a broad absorption band in...Ch. 12 - Prob. 39PCh. 12 - The problem below is directed toward devising a...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Determine the number of protons, neutrons, and electrons in the following atoms: a. a hydrogen atom that has a ...
General, Organic, and Biological Chemistry (3rd Edition)
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
16.159 In qualitative analysis, and are separated from , , and by adding aqueous to a solution that also co...
Chemistry (7th Edition)
Define freezing point.
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
For Practice 1.1
Is each change physical or chemical? Which kind of property (chemical or physical) is demonst...
Principles of Chemistry: A Molecular Approach (3rd Edition)
In qualitative analysis, Ca2+ and Ba2+ are separated from Na+, K+, and Mg2+ by adding aqueous (NH4)2CO3 to a so...
General Chemistry: Atoms First
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the major organic product(s) for the reaction. Use an expanded octet around sulfur to avoid formal charges. H O fuming H₂SO4 Select G → Draw C Rings H More O Erase Q2 Qarrow_forwardно LOH он HCI (aq.) но- G+H +I но но. heat OH ÓMe но но H', CH3OH но- но. K (major product) онarrow_forwardFor the following nitrophenol, draw the resonance structures of the appropriate phenoxide ion to show why the anion is unusually stable OH O,N 4-nitrophenol Draw the molecules on the canvas by choosing buttons from the Tools (for bonds and charges), Atoms, and Templates toolbars.arrow_forward
- How does the following reaction take place, show each step and possible outcomesarrow_forwardNBS ·Br Mg, ether> CO2 9. Determine whether the following reactions are flawed. Put "Yes" on the line for flawed reactions and for reactions that are not flawed (3 points). MeO COME LIAIH4 HgSO4 H3O EtOH H₂O 7 O EtQ OEt 667 MeOarrow_forwardDraw the structures of the starting materials or major products for each of the following reactions (if no reaction is expected to occur, write NO REACTION); H,SO, A + heat он H2/Pd H,SO4 > B heat KMNO4 D HO.arrow_forward
- Draw the major organic product(s) for the reaction. Use an expanded octet around sulfur to avoid formal charges. H fuming H₂SO4 Select Draw Templates More C H S O Erasearrow_forwardPredict the product(s) of the acid/base reactions below. Draw curved arrows to show the formation and breaking of bonds.arrow_forward5. Using curved arrows show the mechanism for the hydrohalogenation for the following reaction. Show how the two possible intermediate products are formed and explain why only one product proceeds to form the product. Clearly indicate attack of electrons and full and partial (8) charges. H-Br Br H-Br Product Intermediate Productsarrow_forward
- 2. Please draw in the arrows that explain each step in the following reactions. Note: you are not responsible for memorizing these reactions. H a) b) d) H + H-CI ·8-8 loh g OCH 3 H + + HOCH 3arrow_forwardWhich of the given reaction schemes would produce the molecule shown below? || ||| II and III I and II HBr ROOR || Br HBr III HBr ROORarrow_forward7. The reaction of methoxide anion with bromoethane to yield the ether ethyl methyl ether and the bromide anon (Br-) is an excellent example of a general reaction type called Sy2 (substitution nucleophilic bimolecular): CH,0+ CH,СH-Br a CH3-0-CH,СH; + Br- a. Change in enthalpy is -103 kJ/mol; the change in entropy is + 0.025 kJ/mol-K. Calculate DG at 300K. b. Is the reaction endergonic or exergonic? c. Is the reaction endothermic or exothermic? d. Use curved arrows to show the complete mechanism. Reaction of 2-methyl-1-butene with H-Cl could yield TWO alkyl chloride products. Draw and name 8. them.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY