
Concept explainers
Convert each molecule into a skeletal structure.
a.
b.
c.
d.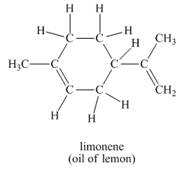
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Organic Chemistry (6th Edition)
Additional Science Textbook Solutions
Chemistry by OpenStax (2015-05-04)
Introduction to Chemistry
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Living By Chemistry: First Edition Textbook
Organic Chemistry (8th Edition)
- Based on the molecular formula, determine whether each compound is an alkane, alkene, or alkyne. (Assume that the hydro- carbons are noncyclical and there is no more than one multiple bond.) a. C5H12 b. C3H6 с. С-Н12 d. C11H22arrow_forwardConvert each condensed formula to a complete structure with lone pairs on heteroatoms. a. CH 3(CH 2) 8CH 3 c. CH 3CCl 3 e. (CH 3) 2CHCH 2NH 2 b. CH 3(CH 2) 4OH d. CH 3(CH 2) 4CH(CH 3) 2arrow_forwardClassify the following hydrocarbons as alkanes, cycloalkane, alkene, cycloalkenes, alkyne, cycloalkyne, or aromatic.a. CH2 CH CH2b. CH3 C (CH3)2 CH (CH2CH3) CH2 CH2 CH (CH3)2c. (CH3)3 C C C CH (CH3) CH2 CH3arrow_forward
- Name each alkane. a. CH3-CH-CH3 ČH; CH; CH3 b. CH3-CH-CH2-CH-CH2 ČH3 CH, CH, c. CH3-C-C-CH3 ČH, ČH3 CH3 CH, CH2 d. CH3-CH-CH2-CH-CH-CH2-CH2-CH3 ČH3arrow_forward1. Identify the circled functional group(s) for each molecule. ОН CH3CHCH3 a. бо =CH₂ b. с. d. NH₂CH₂CH₂CH₂2NHCH3 H₂N 0 CH₂CHCH₂NHCH(CH3)2 стилинен с OHarrow_forward3. Name each of these compounds (including isomer prefixes) and identify the functional groups present. a. H CH CH₂CH/ CH₂ Br b. H CH H El CHSarrow_forward
- what type of isomer? H3C CH3 H. O a. trans O b. E OC.Z O d. cisarrow_forwardBased on the molecular formula, determine whether each compound is an alkane, alkene, or alkyne. (Assume that the hydrocarbons are noncyclic and there is no more than one multiple bond.) a. C,H12 b. CH, c. C;H12 d. CH22arrow_forward1. Which of the following compounds are isomers of each other? I. 3-hexene II. Cyclohexane III. 2,3-dimethyl-2-butene IV. 2-methyl-2,3-pentadiene a. I and II b. II and IV c. I, II, III d. I, II, III, IV 2. Which of the following does not contain a carbonyl group? A. CH3CH2CH2COCH2CH3 B. CH3CH2CH2COOCH3 C. CH3CH2CH2CHOHCH2CH3 D. CH3CH2CH2CONH2CH3arrow_forward
- For each molecule, draw the structure and indicate whether cis/trans isomers are possible. If they do, draw them both and label them as cis and trans. a. 2-methyl-1-butene b.1,2-dimethylcyclohexanearrow_forwardWhich compound is a structural isomer of the following organic compound? ÇH2CH3 CH3CH2-C-CH2CH3 ČH3 Oa. CH3 H3C CH3 H3C ČH3 Ob. CH H,C CH, c. CH2CH3 CH3CH2-C-CH2CH3 ČH3arrow_forwarda. How many Lewis structures have the formula C4H11N? 10 b. In how many of the structures is the nitrogen atom attached to only one carbon? c. How many of them have carbon-carbon double bonds? d. How many of them have carbon-nitrogen double bonds? e. How many of them have rings? f. How many of the structures are capable of hydrogen bonding? g. How many of the structures contain a carbon atom attached to three other carbons? h. How many of the structures contain a nitrogen atom attached to three carbons? Varrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning



