
Organic Chemistry (6th Edition)
6th Edition
ISBN: 9781260119107
Author: Janice Gorzynski Smith
Publisher: McGraw Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1, Problem 43P
Assign formal charges to each
a. 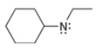 b.
b. 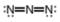 c.
c. 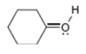 d.
d. 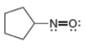
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
The element sulphur has atomic number 16. Sulphur forms a molecular compound with chloride with a molecular formula SCl2. The atomic number of chlorine is 17.
a. Write down the electronic configuration of a sulphur atom and a chlorine atom.
b. Draw a Lewis structure of an SCl2 molecule, by showing the electrons in the outer shells of the atoms.
c. State how many bond pairs and lone pairs of electrons are arranged around the sulphur atoms in SCl2 molecule.
d. Sketch the shape and state the molecular geometry of SCl2 molecule.
e. Explain the hybridisation of the central atom in SCl2 molecule.
f.Explain the intermolecular forces exists in SCl2 molecule.
Draw a Lewis structure for SO2 that obeys the octet rule if possible and answer the following questions based on your drawing.
1. For the central sulfur atom:
- The number of lone pairs = ?
- The number of single bonds = ?
- The number of double bonds = ?
2. The central sulfur atom
a. obeys the octet rule.
b. has an incomplete octet.
c. has an expanded octet.
A. CHF
i. Best Lewis Structure
B. HNO (H is connected to one of the O's)
i. Best Lewis Structure
ii. Electron geometry on the C atom
ii. Electron geometry on the N atom
iii. Approximate bond angles about the C atom
iii. Approximate bond angles around the N atom
v. Draw the shape with in and out wedges (as necessary) and
dipole arrows around the C atom.
v. Draw the shape with in and out wedges (as necessary) and
dipole arrows around the N atom.
vi. Is the molecule polar or nonpolar?
vi. Is the molecule polar or nonpolar?
Chapter 1 Solutions
Organic Chemistry (6th Edition)
Ch. 1.1 - While the most common isotope of nitrogen has a...Ch. 1.2 - Label each bond in the following compounds as...Ch. 1.3 - Draw a valid Lewis structure for each species. a....Ch. 1.3 - Prob. 9PCh. 1.4 - Draw Lewis structures for each molecular formula....Ch. 1.6 - Classify each pair of compounds as isomers or...Ch. 1.6 - Prob. 12PCh. 1.6 - Prob. 13PCh. 1.6 - Prob. 14PCh. 1.6 - Prob. 16P
Ch. 1.6 - Prob. 17PCh. 1.7 - Prob. 18PCh. 1.7 - Prob. 19PCh. 1.7 - Using the principles of VSEPR theory, you can...Ch. 1.8 - Convert each condensed formula to a Lewis...Ch. 1.8 - Prob. 22PCh. 1.8 - Prob. 23PCh. 1.8 - Convert each skeletal structure to a complete...Ch. 1 - Citric acid is responsible for the tartness of...Ch. 1 - Zingerone gives ginger its pungent taste. a.What...Ch. 1 - Assign formal charges to each and atom in the...Ch. 1 - Prob. 44PCh. 1 - Prob. 46PCh. 1 - Draw all possible isomers for each molecular...Ch. 1 - 1.45 Draw Lewis structures for the nine isomers...Ch. 1 - Prob. 52PCh. 1 - Prob. 53PCh. 1 - Prob. 54PCh. 1 - Consider compounds A-D, which contain both a...Ch. 1 - Draw in all the carbon and hydrogen atoms in each...Ch. 1 - 1.61 Convert each molecule into a skeletal...Ch. 1 - Prob. 65PCh. 1 - Predict the hybridization and geometry around each...Ch. 1 - Prob. 68PCh. 1 - Ketene, , is an unusual organic molecule that has...Ch. 1 - Rank the following bonds in order of increasing...Ch. 1 - Two useful organic compounds that contain Cl atoms...Ch. 1 - Use the symbols + and to indicate the polarity of...Ch. 1 - Prob. 74PCh. 1 - Anacin is an over-the-counter pain reliever that...Ch. 1 - 1.77 Stalevo is the trade name for a medication...Ch. 1 - 1.78 and are two highly reactive carbon...Ch. 1 - 1.79 The N atom in (acetamide) is hybridized,...Ch. 1 - Prob. 83PCh. 1 - Prob. 84PCh. 1 - Prob. 85PCh. 1 - Prob. 86PCh. 1 - Prob. 87PCh. 1 - Prob. 88P
Additional Science Textbook Solutions
Find more solutions based on key concepts
Give one example from main group chemistry that illustrates each of the following descriptions: (a) Covalent ne...
General Chemistry: Atoms First
Practice Problem 1.22 Which of the following alkenes can exist as cis-trans isomers? Write their structures. Bu...
Organic Chemistry
The smallest building blocks inside your cell phone are about 1000 times smaller than the diameter of a human h...
Chemistry In Context
141. Design a device that uses as electrochemical cell to determine amount of
in a sample water Describe, in...
Chemistry: Structure and Properties (2nd Edition)
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw in all the hydrogen atoms and nonbonded electron pairs in each ion. a. b. d. ENHarrow_forwardWhich bond in each pair is more polar—that is, has the larger electronegativity difference between atoms? a. C- O or C-N b. C-F or C-Cl c. Si-C or P-Harrow_forwardd. H E Assign formal charges to each carbon atom in the given species. All lone pairs have been drawn in. H H a. CH2-CH b. H-C-H c. H-C-H ннarrow_forward
- To answer the questions, interpret the following Lewis diagram for SO42- . 1. For the central sulfur atom: ... The number of non-bonding electrons = The number of bonding electrons = The total number of electrons = 2. The central sulfur atom fill in the blank 4 ... A. obeys the octet rule. B. has more than an octet. C. has less than an octet.arrow_forwardDraw a Lewis structure for BF3 that obeys the octet rule if possible and answer the following questions based on your drawing. 1. For the central boron atom: - The number of lone pairs = ? - The number of single bonds = ? - The number of double bonds = ? 2. The central boron atom is a. obeys the octet rule. b. has an incomplete octet. c. has an expanded octet.arrow_forwardDraw a resonance structure that places a pi bond in a different position. Include all lone pairs in your structure. :O: H. H. 工 :O:arrow_forward
- Carbon ring structures are common in organic chemistry. Draw a Lewis structure for each carbon ring structure, including any necessary resonance structures. a. CHg b. CH4 c. CH12 d. C,H.arrow_forward9. Indicate whether each statement below is true or false. a. The core electrons are called valence electrons. b. lonic bonds are formed by the attraction between cations and anions. c. Ionic bonds are formed from atoms by a transfer of electrons. d. In a Lewis symbol, the chemical symbol represents the nucleus of the atom. e. An ion written as a Lewis symbol has brackets outside the electrons. f. Two electrons involved in a bond produce a double bond. g. If the AEN value is very large the bond is polar covalent. h. The central atom is typically the atom with the highest electronegativity. i. An expanded octet has larger electron clouds. j. A nonpolar molecule can have polar bonds.arrow_forwardDraw in all the hydrogen atoms and nonbonded electron pairs in each ion. a. b. с. d.arrow_forward
- Draw the Lewis structure for bromoethane (C,H,Br). Be certain you include any lone pairs. C. C.arrow_forwardDraw the Lewis structure for a ClO- ion on a separate piece of paper. a. How many lone pairs of electrons are of the Cl atom? b. How many lone pairs of electrons are on the O atom? c. Is the bond between chlorine and oxygen a single, double, or triple bond?arrow_forwardDetermine if a bond between each pair of atoms would be pure covalent, polar covalent, or ionic.a. Br and Br b. C and Clc. C and S d. Sr and Oarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY