
Organic Chemistry-Package(Custom)
4th Edition
ISBN: 9781259141089
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1, Problem 1.80P
The principles of this chapter can be applied to organic molecules of any size. Answer the following questions about amoxicillin, an antibiotic from the penicillin family.
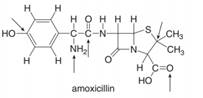
a. Predict the hybridization and geometry d. How many
around each indicated atom. Label them.
b. Label five polar bonds using the e. Find a
symbols
c. Draw the skeletal structure.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Answer the following questions about amoxicillin, an antibiotic from the penicillin family.
a. Predict the hybridization and geometry around each highlighted atom.
b. Label five polar bonds using the symbols δ+ and δ–.
c. How many π bonds does amoxicillin have? Label them.
d. Find a C – H bond containing a carbon atom having a hybrid orbital with 33% s-character
B. Answer the next two questions about the Lewis structure
of XYZ shown below.
メー三
-YEz:
I. What is the hybridization of the central atom?
II. For all covalent bonds, specify the type of bond (o vs. T)
and the atomic orbitals that overlap to form the bond.
For example, in a molecule of H2, the only bond is o:
H(s)-H(s).
сс с
C-C= C
C-C C- C C
a. Complete the formula, number the carbon atoms and name the molecule
b. Draw and label in terms of the overlapping hybrid orbitals forming each bond
c. Identify the hybrid orbitals forming each bond
For your answers for letters d,e,f, give the number of the C atom (from a) only (ex. C10 -
C11)
d. Which C to C bond is the strongest
e. Which C to H bond is the most polar
f. Which C to C bond is the least free to rotate
g. What is the mobile bond order
Chapter 1 Solutions
Organic Chemistry-Package(Custom)
Ch. 1 - While the most common isotope of nitrogen has a...Ch. 1 - Label each bond in the following compounds as...Ch. 1 - How many covalent bonds are predicted for each...Ch. 1 - Draw a valid Lewis structure for each species. a....Ch. 1 - Draw an acceptable Lewis structure for each...Ch. 1 - Prob. 1.6PCh. 1 - Draw a Lewis structure for each ion. a. CH3Ob....Ch. 1 - Draw Lewis structures for each molecular formula....Ch. 1 - Prob. 1.9PCh. 1 - Prob. 1.10P
Ch. 1 - Prob. 1.11PCh. 1 - Prob. 1.12PCh. 1 - Draw a second resonance structure for each...Ch. 1 - Prob. 1.14PCh. 1 - Draw a second resonance structure for nitrous...Ch. 1 - Prob. 1.16PCh. 1 - Prob. 1.17PCh. 1 - Prob. 1.18PCh. 1 - Prob. 1.19PCh. 1 - Prob. 1.20PCh. 1 - Simplify each condensed structure by using...Ch. 1 - Prob. 1.22PCh. 1 - Prob. 1.23PCh. 1 - Convert each skeletal structure to a complete...Ch. 1 - Draw in all hydrogens and lone pairs on the...Ch. 1 - Prob. 1.26PCh. 1 - What orbitals are used to form each of the CC, and...Ch. 1 - What orbitals are used to form each bond in the...Ch. 1 - Determine the hybridization around the highlighted...Ch. 1 - Classify each bond in the following molecules as ...Ch. 1 - Prob. 1.31PCh. 1 - Rank the following atoms in order of increasing...Ch. 1 - Prob. 1.33PCh. 1 - Prob. 1.34PCh. 1 - Provide the following information about...Ch. 1 - Use the ball-and-stick model to answer each...Ch. 1 - Citric acid is responsible for the tartness of...Ch. 1 - Zingerone gives ginger its pungent taste. a.What...Ch. 1 - Two radioactive isotopes of iodine used for the...Ch. 1 - Prob. 1.40PCh. 1 - Assign formal charges to each carbon atom in the...Ch. 1 - Assign formal charges to each N and O atom in the...Ch. 1 - Draw one valid Lewis structure for each compound....Ch. 1 - Prob. 1.44PCh. 1 - Prob. 1.45PCh. 1 - Prob. 1.46PCh. 1 - Draw all possible isomers for each molecular...Ch. 1 - 1.45 Draw Lewis structures for the nine isomers...Ch. 1 - Prob. 1.49PCh. 1 - Prob. 1.50PCh. 1 - Prob. 1.51PCh. 1 - Prob. 1.52PCh. 1 - Prob. 1.53PCh. 1 - Prob. 1.54PCh. 1 - Draw all reasonable resonance structures for each...Ch. 1 - Prob. 1.56PCh. 1 - Rank the resonance structures in each group in...Ch. 1 - 1.56 Consider the compounds and ions with curved...Ch. 1 - 1.57 Predict all bond angles in each...Ch. 1 - Predict the geometry around each indicated atom....Ch. 1 - Prob. 1.61PCh. 1 - Prob. 1.62PCh. 1 - Draw in all the carbon and hydrogen atoms in each...Ch. 1 - Prob. 1.64PCh. 1 - Prob. 1.65PCh. 1 - Prob. 1.66PCh. 1 - Prob. 1.67PCh. 1 - Each of the following condensed or skeletal...Ch. 1 - Prob. 1.69PCh. 1 - Prob. 1.70PCh. 1 - Prob. 1.71PCh. 1 - Prob. 1.72PCh. 1 - Prob. 1.73PCh. 1 - Prob. 1.74PCh. 1 - Two useful organic compounds that contain Cl atoms...Ch. 1 - Use the symbols + and to indicate the polarity of...Ch. 1 - Label the polar bonds in each molecule. Indicate...Ch. 1 - Answer the following questions about acetonitrile...Ch. 1 - Prob. 1.79PCh. 1 - The principles of this chapter can be applied to...Ch. 1 -
a. What is the hybridization of each N atom in...Ch. 1 - 1.77 Stalevo is the trade name for a medication...Ch. 1 - 1.78 and are two highly reactive carbon...Ch. 1 - 1.79 The N atom in (acetamide) is hybridized,...Ch. 1 - Prob. 1.85PCh. 1 - Prob. 1.86PCh. 1 - Prob. 1.87PCh. 1 - Prob. 1.88PCh. 1 - Prob. 1.89PCh. 1 - Prob. 1.90P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A molecular compound is composed of 58.8% Xe, 7.2%. O, and 34.0%. F, by mass. If the molecular weight is 223 amu, what is the molecular formula? What is the Lewis formula? Predict the molecular geometry using the VSEPR model.arrow_forwardPredict the valence electron molecular orbital configurations for the following, and state whether they will be stable or unstable ions. (a) Na,2+ (b) Mg,2 (c) AI,2 (d) Si,2 (e) p2+ (f) s,2 (g) F,2 (h) Ar,2 40. Predict the valence electron molecular orbital configurations for the following, and state whether they will be stable or unstable ions. (a) Na22+ (b) Mg22+ (c) Al22+ (d) Si22+ (e) P22+ (f) S22+ (g) F22+ (h) Ar22+arrow_forwardConsider three molecules: A, B, and C. Molecule A has a hybridization of sp3 Molecule B has two more effective pairs (electron pairs around the central atom) than molecule A. Molecule C consists of two bonds and two bonds. Give the molecular structure, hybridization, bond angles, and an example for each molecule.arrow_forward
- For each of the following molecules, state the bond angle (or bond angles, as appropriate) that you would expect to see on the central atom based on the simple VSEPR model. Would you expect the actual bond angles to be greater or less than this? a CCl4 b SCl2 c COCl2 d AsH3arrow_forwardIn Chapter 6, we study a group of organic cations called carbocations. Following is the structure of one such carbocation, the tert-butyl cation. (a) How many electrons are in the valence shell of the carbon bearing the positive charge? (b) Using VSEPR, predict the bond angles about this carbon. (c) Given the bond angle you predicted in (b), what hybridization do you predict for this carbon?arrow_forwardAnswer the following questions about amoxicillin, an antibiotic from the penicillin family. NH2 H N. N. НО Но amoxicillin a. Predict the hybridization and geometry around each highlighted atom. b. Label five polar bonds using the symbols &+ and 8–. C. How many n bonds does amoxicillin have? Label them. d. Find a C- H bond containing a carbon atom having a hybrid orbital with 33% s-character.arrow_forward
- Consider NH3 and PH3. Electronegativities: P = 2.1, H = 2.1, N=3.0. Which statement is false? O A. The P-H bonds are more polar than the N-H bonds. OB. The bond dipoles in NH3 are directed toward the nitrogen atom. For PH3 there is no bond dipole moment. OC. Both are sp hybridized at the central atom. に O D. There are no false statements. All of these statements are corect. OE. Both molecules are polar.arrow_forward8. Consider the following organic molecules. What are the hybridizations around each carbon atom? What are the shapes around each carbon atom? You may want to number your carbon atoms for clarity. H H H H. C-H .C H. H-C=C-H C H C= H.arrow_forward9. Show the dipole moment of each bond and then predict the overall molecule as either being polar or nonpolar? a. CH4 b. H2S c. PH3 d. SO3arrow_forward
- 1. Ethanol, C,H,0 (CH,CH,OH) Lewis structure using dots to represent bonding electrons: Lewis structure using lines to represent pairs of bonding electrons: Label the carbon bonded to only hydrogen and other carbon atom as carbon A, and the other carbon atom as carbon B. Carbon atom A: Total number of electron groups: Number of lone pairs: Number of bonding groups/pairs: Bond angles: Electron geometry: Molecular geometry: Hybridization: Number of hybrid orbitals: Carbon atom B: Total number of electron groups: Number of lone pairs: Number of bonding groups/pairs: Bond angles: Electron geometry: Molecular geometry: Hybridization: Number of hybrid orbitals: Oxygen atom Total number of electron groups: Number of lone pairs: Number of bonding groups/pairs: Bond angles: Electron geometry: Molecular geometry: Hybridization: Number of hybrid orbitals: Is this molecule polar? (Y/N) Can this molecule hydrogen bond with itself? Number of sigma bonds: Number of pi bonds: MacBook Proarrow_forwardFill in each blank sentence with numbers 0-10. An sp3-hybridized atom possess _____ s orbital(s), _______ p orbital(s), and _____ hybrid orbital(s) in its valence shell. Such an atom can form up to _____ sigma bonds and _____ pi bonds.arrow_forwardWhat are the angles a and b in the actual molecule of which this is a Lewis structure? H- H a-0° b = 1⁰ C H a b H Note for advanced students: give the ideal angles, and don't worry about small differences from the ideal that might be caused by the fact that different electron groups may have slightly different sizes. X Sarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY