
Concept explainers
(a)
Interpretation: The orbitals used to form the indicated bonds are to be predicted.
Concept introduction: The geometry and hybridisation of an atom is determined by the number of group around it. If the number of groups attached to an atom is
Answer to Problem 1.70P
Orbitals used in the formation of given bonds are:
Explanation of Solution
The given compound is,
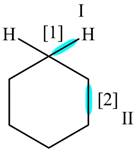
Figure
In bond
Orbitals used in the formation of given bonds are:
(b)
Interpretation: The orbitals used to form the indicated bondsare to be predicted.
Concept introduction: The geometry and hybridisation of an atom is determined by the number of group around it. If the number of groups attached to an atom is
Answer to Problem 1.70P
Orbitals used in the formation of given bonds are:
Explanation of Solution
The given compound is,
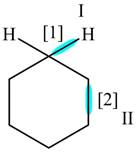
Figure
In bond
Orbitals used in the formation of given bonds are:
(c)
Interpretation: The orbitals used to form the indicated bonds are to be predicted.
Concept introduction: The geometry and hybridisation of an atom is determined by the number of group around it. If the number of groups attached to an atom is
Answer to Problem 1.70P
Orbitals used in the formation of given bonds are:
Explanation of Solution
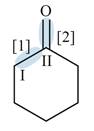
Figure 2
In bond
Orbitals used in the formation of given bonds are:
(d)
Interpretation: The orbitals used to form the indicated bonds are to be predicted.
Concept introduction: The geometry and hybridisation of an atom is determined by the number of group around it. If the number of groups attached to an atom is
Answer to Problem 1.70P
Orbitals used in the formation of given bonds are:
Explanation of Solution
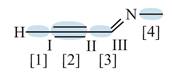
Figure 3
In bond
Orbitals used in the formation of given bonds are:
Want to see more full solutions like this?
Chapter 1 Solutions
Organic Chemistry-Package(Custom)
- Write a dash, condensed and bond-line structural formulas of each compound given below. Structural Formula Ball-and-stick Model Dash Condensed Bond-line a. b. C. d. *Note: White = H, Black = C and Red = Oarrow_forwardThe two carbon atoms in acetylene form two pi bonds. O A. True, hence a double bond O B. True, and two sigma bonds C. False, no double bond present D. False, two sigma bonds onlyarrow_forwardDraw a second resonance structure for each ion. Then, draw the resonance hybrid. :0: :0: :OH а. b. C. :ö:arrow_forward
- Consider a molecule of methanimine (CH3N) compared to a molecule of methylamine (CH5N). The C-N bond in methanimine is than the C-N bond in methylamine. A. longer and stronger B. longer and weaker C. shorter and stronger D. shorter and weaker E. the same strength and length asarrow_forwardWhat orbitals are used to form each highlighted bond? For multiple bonds, indicate the orbitals used in individual bonds.arrow_forwardd. b. e o C. a. O 6. Draw at least 3 additional (l.e., in addition to the structure provided) valid resonance (major contributors only) forms of the following ions: Ⓒarrow_forward
- With reference to compound A drawn below, label each compound as an isomer, a resonance structure, or neither. :0: :0: :O: :ö: :0: a. b. с. d. A With reference to cation B, label each species as an isomer, a resonance structure, or neither. а. b. С. d.arrow_forwardAnswer the following questions about octocrylene, a common sunscreen component. a. What is the hybridization of each C atom? b. How many lone pairs does octocrylene contain? c. What is the geometry around each C atom? d. Draw two additional resonance structures. e. Label all polar bonds.arrow_forwardFollow the curved arrows and draw a second resonance structure for each species. a. CH,-NEN: b. CH3-C-CH CHe NH2 C. d. CHarrow_forward
- Draw all reasonable resonance structures for each species. a. O3 c. Ng e. g. :O: :O: b. NO, (a central N atom) d. CH3-C-CH-C-CH, 1. CH2=CH-CH-CH=CH2arrow_forwardConsider tne pairs oI SIruciures snown. H. H. А. and H. CH3 CH3 В. and H. :O: and H3C H. H2C D. CH3CH,* and *CH2CH3 Which structures are resonance structures to each other? pair A pair B pair C nair D :O: C.arrow_forwardMCQ 73: The physical properties of bonding are influenced by bonding between A. atoms B. ions C. molecules D. all of abovearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





